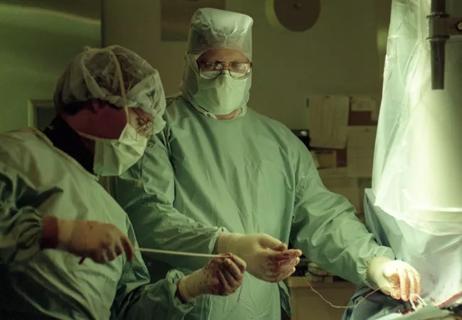
A pioneer of CIED lead extraction shares wisdom in HRS Founders Lecture
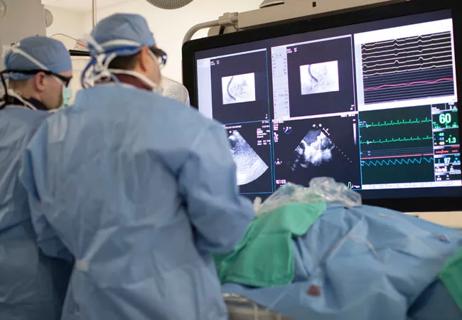
The key: Linking research with practice to tackle complex cases

Lead investigator shares registry highlights, implications
LEADconnection.org forges a global community to advance best practice
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy

From leadless pacing to drugging the microbiome for atherosclerosis

From prevention and pacing to stenting and surgery

Exciting advances are improving outcomes and safety
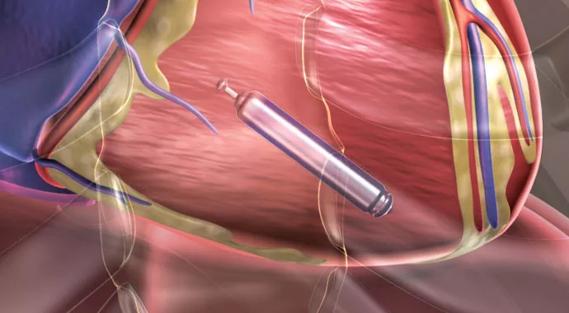
With launch of U.S. trials, the holy grail of leadless pacing seems near
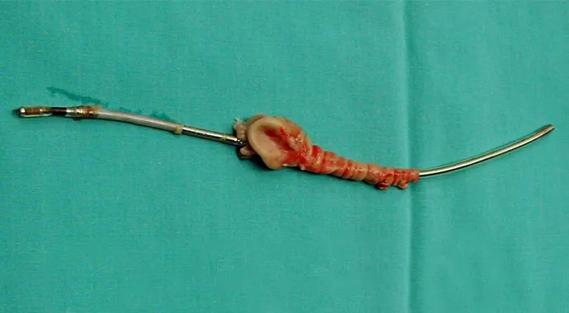
Removing faulty or infected device leads is complex — and growing more common

System alerts to problems, stores data for research