Key step in nation’s first clinical trial of transplant for uterine infertility
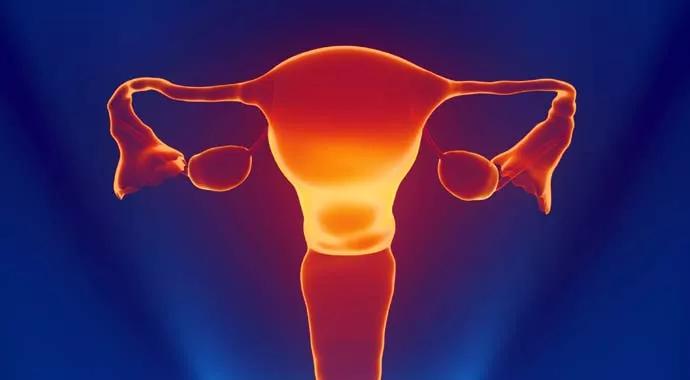
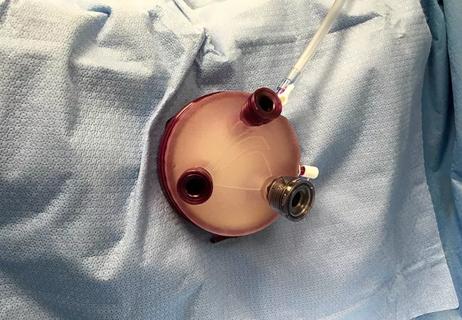
A multidisciplinary Cleveland Clinic surgical team performed the nation’s first uterine transplant during a nine-hour surgery Feb. 24, 2016. The procedure was the first in a pioneering Cleveland Clinic trial of uterine transplantation in women of reproductive age with uterine factor infertility (UFI).
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
The IRB-approved study — the first of its kind in the United States and one of the first in the world — is spearheaded by Cleveland Clinic transplant surgeon Andreas Tzakis, MD, PhD.
“We are very excited. Uterine transplantation presents a unique option for women coping with UFI,” says Dr. Tzakis. “At the same time, we are aware of the tremendous responsibility we have undertaken. This is a complex trial, and we must make sure everything we do gives our patients the best opportunity for a normal delivery.”
Dr. Tzakis, who has done independent research on uterine transplantation, has been involved in an ongoing uterine transplantation trial with colleagues at the University of Gothenburg, Sweden, for years. The Swedish team has performed several uterine transplants to date. At least seven of them have yielded viable uteri with menses, resulting in five live births so far. The first live birth was reported in The Lancet in early 2015.
Tommaso Falcone, MD, chairman of Cleveland Clinic’s Ob/Gyn & Women’s Health Institute, has followed the Swedish trial closely. He is an active investigator in the field of infertility and reproductive surgery and has been interested in uterine transplantation for more than a decade.
“Adoption or maternal surrogacy are not always acceptable options for social, ethical, cultural or religious reasons,” says Dr. Falcone. “Uterine transplantation is an important development that will give these women the opportunity to have a biological child.”
Uterine transplantation differs from other organ transplants in one key aspect: Because its benefit ceases after childbearing is complete, the organ is removed or allowed to be rejected after the baby or babies are born. This eliminates the need for lifelong immunosuppression and associated medical problems.
The prospect of an “ephemeral” transplant has raised ethical issues surrounding the procedure and the safety of the transplant recipients, as well as the babies born of the process. For this reason, Cleveland Clinic bioethicists were intimately involved in discussions leading to IRB approval of the clinical trial and will remain involved as the trial progresses.
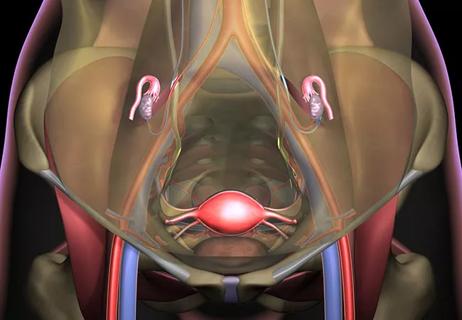
Ten women ages 21 to 39, with infertility due to congenital uterine absence (Mayer-Rokitansky syndrome) or because their uterus was removed, will be selected to receive a transplant from a deceased donor.
The Swedish study uses living donors. However, Cleveland Clinic is pursuing only deceased donor organs to avoid any risk to living donors.
Lifebanc will identify deceased donors of menstruating age with the same blood type and a healthy uterus.
The multi-phase trial begins with a detailed history and physical, a fertility assessment by a reproductive gynecologist, a psychiatric review, a discussion with a bioethicist and a review of social support.
“This is a lengthy evaluation process designed to ensure each woman has the personality, social support and other circumstances that will allow a successful transplant,” says John Fung, MD, PhD, Director of the Cleveland Clinic’s Health System Center for Transplantation.
A medical examination follows. All criteria must be met in order to be accepted into the study.
Each woman then undergoes ovarian stimulation, egg retrieval and in vitro fertilization with sperm in the laboratory. Once 6 to 10 viable embryos have been frozen, the woman is put on a transplant list to await a suitable donor.
After assurance that the transplant has been successful and that the uterus is healthy and viable — which takes about 12 months — a fertilized embryo will be implanted. The patient’s frozen embryos will be implanted one at a time as needed until one to two healthy births are achieved.
“We want to make sure the uterus is mature and stable to give the embryo the best possibility for success,” says Rebecca Flyckt, MD, Director of the Fertility Preservation Program in the Ob/Gyn & Women’s Health Institute. “We also think this timetable is safest for the recipient.”
If an embryo results in pregnancy, the patient will be followed by Cleveland Clinic maternal/fetal medicine specialists, who are accustomed to caring for high-risk pregnancies. Delivery will be made by caesarean section. After birth, the baby will be followed by advanced neonatologists.
With an experienced transplant group and a team of adult, pediatric, neonatal and obstetric specialists accustomed to managing high-risk, complex patients, Cleveland Clinic is ideally suited to undertake uterine transplantation.
The complexity of the clinical trial requires a team of specialists very different from the one involved in a typical organ transplant, and includes:
Also on the team are Deb Plavney, Deborah Sears, RN, Janelle Romond, Trudy Aquilina, LISW-S, CCTSW, Terrence Roncagli, LISW, Susan Thomas, Megan Villarreal and Stacy Payne.
Treatment being offered in cases where medical and hormonal management was not successful
Standardizing obstetrics and gynecology education while promoting patient safety

High physician/patient convenience and satisfaction cited

Automated visual evaluation of the cervix
Don’t miss this educational opportunity
The next evolution in MIS is coming sooner than you think
Limit radiation damage to ovaries without removing them
Trainees no longer need to “practice” on patients