Worries about cerebral blood flow & brain oxygenation unfounded

By Ehab Farag, MD, FRCA, and Michael Kot, MD
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
Dexmedetomidine is considered a nearly ideal sedative drug in the perioperative period and in most ICU settings. It maintains normal sleep patterns, limits impaired ventilation and maintains good patient responsiveness during conscious sedation. It has also been shown to reduce incidence of delirium in the postoperative period. Because the drug reduces cerebral blood flow without reducing cerebral metabolic rate of oxygen, its safety in patients with neurological diseases is uncertain. Thus, dexmedetomidine is not widely used in neurointensive care settings. Our team hypothesized that this worry is unfounded, and confirmed in a randomized noninferiority trial that dexmedetomidine is also an excellent sedative choice for patients with neurodegenerative disorders.
Our study tested the hypothesis that dexmedetomidine was noninferior to propofol in regards to cerebral blood flow rate and brain oxygenation on 44 patients scheduled for insertion of deep-brain stimulating electrodes and then randomized to either dexmedetomidine (n = 23) or propofol (n = 21). We used transcranial Doppler (TCD) to measure the cerebral blood flow velocities as surrogate to cerebral blood flow, and near infrared spectrospy (NIRS) to measure the brain oxygenation in deep brain stimulator insertion (DBS) procedures for patients with neurodegenerative disorders like Parkinson’s disease and essential tremors.
The barred use of opioids and other sedatives like benzodiazepines during DBS procedures makes it an ideal model to test only the effects of dexmedetomidine or propofol on cerebral blood flow and oxygenation without the influence of other drugs.
The results of our study show that dexmedetomidine is noninferior to propofol with regards to cerebral blood flow and brain oxygenation in patients with Parkinson’s disease and tremors. It also showed that dexmedetomidine provided deeper sedation without significant differences in the number of hypertensive or apneic episodes, pulsatility index or cerebral profusion pressure.
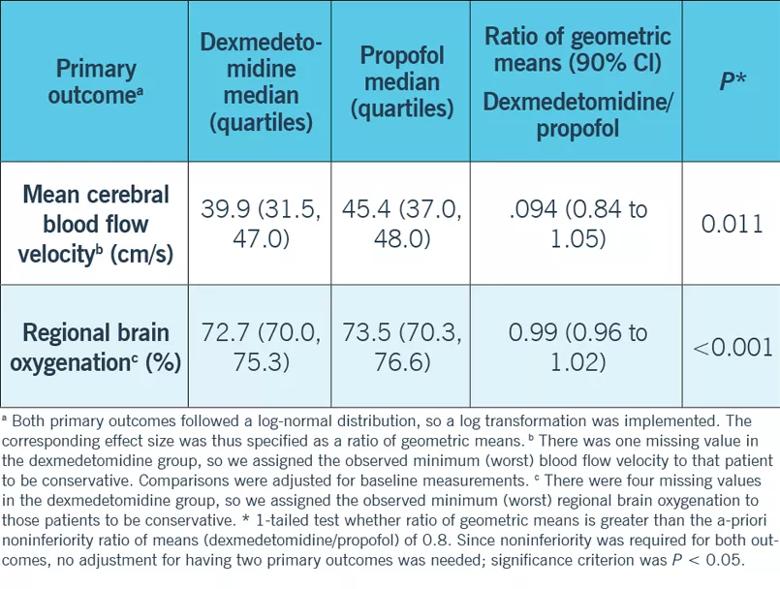
As the clinical indications for DBS widen, the advantages of dexmedetomidine over other sedatives reach more and more patients. We hope that this study paves the way for further, larger studies of using dexmedetomidine in patients with other neurological diseases and in neurointensive care patients.
Dr. Farag is Director of Clinical Research in the Department of General Anesthesiology at Cleveland Clinic and Professor of Anesthesiology at Cleveland Clinic Lerner College of Medicine. Dr. Kot is a fellow in Critical Care Medicine.

Program enhances cooperation between traditional and non-pharmacologic care
Pain specialists can play a role in identifying surgical candidates

Clinical judgment is foundational to appropriately prescribing
The device is a less invasive alternative for patients who are struggle with chronic pain

The Four Pillar Project provides physicians with evidence-based talking points to quickly identify problem areas and educate patients to optimize important aspects of their health.
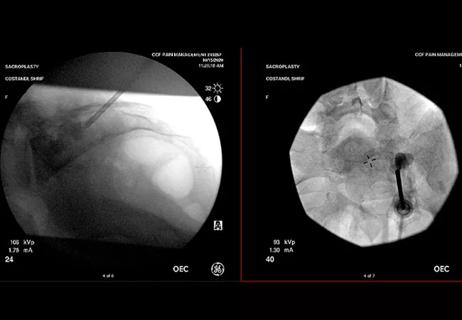
When conservative approaches don’t work, it may be time to turn to sacroplasty
Fourth iteration of the guidelines focuses on high-risk patient identification and evidence on newer drugs

It is a lifestyle commitment that can minimize pain and improve lives