Cleveland Clinic is part of a multicenter Phase 3 clinical trial comparing the effect of adding cabozantinib to avelumab versus avelumab alone in patients with metastatic urothelial cancer who responded to first-line platinum-based chemotherapy. The trial, dubbed MAIN-CAV, is led by the Alliance for Clinical Trials in Oncology and formally opened in March 2022. The trial is currently active and enrolling at the Cleveland Clinic, and aims to enroll 650 adult participants across the US and Canada.
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
“In urothelial cancer, the combination of chemotherapy and immunotherapy in the front-line setting has not shown beneficial compared to just chemotherapy, but sequential use with maintenance immunotherapy has shown benefit,” says National Principal Investigator Shilpa Gupta, MD of the Taussig Cancer Center at Cleveland Clinic. “In the JAVELIN Bladder 100 trial, the addition of avelumab to the best supportive care improved survival by seven months. In the MAIN-CAV trial, we are pushing the envelope further by comparing avelumab alone to the combination of avelumab and the targeted therapy cabozantinib to determine if we can improve the overall survival benefit seen with maintenance avelumab by another seven months.”
The trial will enroll only patients who have achieved stable disease, or partial or complete responses after first-line platinum-based chemotherapy.
Avelumab is a PD-L1 inhibitor approved by the FDA in June of 2020 as maintenance therapy for patients with locally advanced or metastatic urothelial carcinoma that has not progressed with first-line platinum-based chemotherapy.
Cabozatinib is an oral vascular endothelial growth factor (VEGF) tyrosine kinase inhibitor (TKI) approved for the treatment of advanced renal cell carcinoma, hepatocellular carcinoma and differentiated thyroid cancer. The analogy behind combining avelumab with cabozatinib in patients with metastatic urothelial cancer is to harness the power of two synergistic agents with different mechanisms of action in the hope of maintaining a longer-term response in the maintenance setting.
In previous preclinical and clinical studies, synergy has been observed between cabozatinib and other immunotherapies, such as nivolumab and etazolizumab in urothelial cancer with around 20% response rates.
“Based on the data from urothelial cancer studies, cabozatinib partners well with immunotherapy,” Dr. Gupta says. “Combining cabozatinib with immunotherapy can potentially overcome resistance to immunotherapy, because not everyone benefits from single-agent immunotherapy and a lot of people progress on it. Our goal is to maximize the response and the durability of response to immunotherapy [by adding cabozatinib].’’
The MAIN CAV trial is the first in the field trial looking at intensification of maintenance avelumab. If study findings turn out to be positive, Dr. Gupta is hopeful that the avelumab plus cabozatinib combination may potentially become the new standard of care for patients with metastatic urothelial cancer in the maintenance setting.” Exploratory biomarker work from this trial will also help us understand mechanisms of resistance to immunotherapy and which patients will benefit most from combination approaches”.

First-of-its-kind research investigates the viability of standard screening to reduce the burden of late-stage cancer diagnoses
Study demonstrates ability to reduce patients’ reliance on phlebotomies to stabilize hematocrit levels

Findings highlight an association between obesity and an increased incidence of moderate-severe disease
Cleveland Clinic Cancer Institute takes multi-faceted approach to increasing clinical trial access
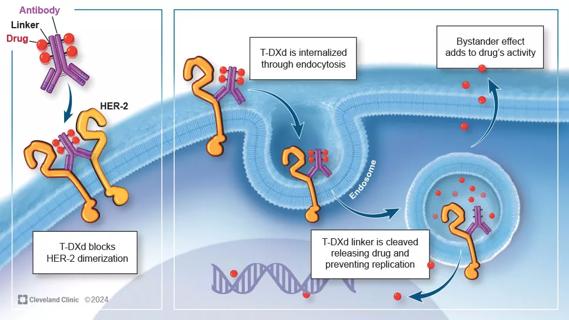
Key learnings from DESTINY trials

Gene editing technology offers promise for treating multiple myeloma and other hematologic malignancies, as well as solid tumors

Study of 401,576 patients reveals differences in cancer burdens as well as overall survival

Enfortumab plus pembrolizumab reduced risk of death by 53% compared with platinum-based chemotherapy