Enrollment is now open for the pioneering precision medicine clinical trial known as the National Cancer Institute-Molecular Analysis for Therapy Choice (NCI-MATCH). Approximately 1,000 patients with advanced solid tumors or lymphomas that have not responded to standard therapy may find new treatment options through this study.
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
This phase II, interventional trial is enrolling patients at clinical sites participating in the NCI’s National Clinical Trials Network (NCTN), including Cleveland Clinic.
In this trial – the largest precision oncology trial ever launched, according to the National Cancer Institute – researchers will analyze each patient’s tumor for the presence of genomic abnormalities for which a targeted drug exists. If a match is found, treatment may be assigned.
“This trial is a well-designed attempt to determine whether cancers can be treated according to their molecular profiles, following the premise of personalized medicine,” says Petros Grivas, MD, PhD, of Cleveland Clinic’s Department of Hematology and Medical Oncology. Dr. Grivas is a national Principal Investigator for one of the trial’s 20 treatment arms, and thus is helping to develop a treatment subprotocol.
The phase II interventional trial, known as the National Cancer Institute-Molecular Analysis for Therapy Choice (NCI-MATCH), is enrolling patients at as many as 2,400 clinical sites that participate in the NCI’s National Clinical Trials Network (NCTN). Cleveland Clinic is working to activate the trial as soon as it becomes available as a member of that network.
The trial will investigate an evolving approach to cancer treatment. “With these new therapies we are targeting the biology of the cancer, not the organ in which it is detected, with agents designed to target that biology,” says Matt Kalaycio, MD, Chairman of the Department of Hematologic Oncology and Blood Disorders at Cleveland Clinic’s Taussig Cancer Institute.
“This trial is a well-designed attempt to determine whether cancers can be treated according to their molecular profiles…”
This approach is designed to better target patients who may benefit, while sparing potential toxicity to those who may not gain from a particular treatment, Dr. Grivas says. “This trial is certainly paradigm-changing, since it moves away from the conventional histology-based eligibility to adopt a molecular-based strategy for patient allocation to relevant treatment arms. The premise is that genetic alterations can act as biomarkers predictive of response to specific targeted agents.”
Enrollment is open to adults with advanced solid tumors, including rare tumors, and lymphomas, that did not respond (or are no longer responding) to standard treatment, or for which no standard therapy exists.
Patients will be screened with a tumor biopsy and the specimens will be sent to one of four standardized genomic testing labs to be analyzed for more than 4,000 different variants across 143 genes. The turnaround time for genomic sequencing is expected to be between 11 and 14 days.
If a tumor is found to have a genomic abnormality that may respond to one or more targeted drugs, the patient may be eligible and will be screened for treatment. Patients with tumors that share the same genomic abnormality, regardless of tumor type, will receive the drug that targets that abnormality.
For example, physicians may prescribe the same drug therapy for a patient with prostate cancer and another patient with bladder cancer if they exhibit the same genomic alteration. Treatment may continue, as per standard clinical research practice, until the disease progresses or unacceptable toxicity levels are reached. However, not all patients will have tumors with an abnormality that matches a drug being tested.
Drugs included in the NCI-MATCH trial have been approved by the U.S. Food and Drug Administration (FDA) for use in treating cancers other than the current application, or are still being tested but have shown some effectiveness against tumors with one or more specific genomic alteration.
Different drugs will be tested in separate arms of the trial. The list of drugs includes crizotinib, dabrafenib, trametinib, sunitinib, afatinib, ado-trastuzumab emtansine, and several other targeted agents. NCI-MATCH may add or drop treatments over the course of the study.
The trial’s primary outcome measure is objective response rate (ORR), defined as the percentage of patients whose tumors have complete or partial response to treatment. “Promising” treatments worthy of further study are those that result in more frequent tumor shrinkage compared to what is expected based on historical control rates. . A secondary endpoint is six-month progression-free survival rate. The trial also will evaluate the time between treatment initiation and cancer progression or death, and treatment side effects.
NCI-MATCH’s broad, inclusive design is because there are many potential molecules to be tested, and many different mutations and targets, Dr. Kalaycio says. “Doing individual trials for each molecule would be cumbersome, expensive and impractical, and by creating this trial design, we get to the answers for some of these agents much faster.”
Dr. Kalaycio says he expects “umbrella” or “basket- type” targeted studies similar to NCI-MATCH to become the norm in the near future, as opposed to large traditional studies of patients with a given cancer, treated with a combination of chemotherapies, which look for outcomes of interest such as remission rates.
He cautions that most patients who sign up for the NCI-MATCH trial’s tumor biopsies will not receive targeted therapy, because their tumors will not have a lesion amenable to treatment by one of the trial’s agents.
“We anticipate that one or two out of every 10 patients we screen will be eligible to actually go on the trial,” he said. Additionally, at least 25 percent of the slots have been reserved for rare tumors, meaning that the trial could be closed to common cancers such as breast and lung much sooner than for those cases where cancer rarely occurs, such as in the eye, ureter and pituitary gland.
Dr. Grivas says it is difficult to estimate how much time it will take for the study to get to full accrual. However, it is anticipated that while some treatment arms may close, others may open – which is another innovative aspect of this trial, he says.
“The infrastructure, logistics and design will remain the same, but new treatment arms may activate over time; the exact total study duration remains unknown at present,” he says.
It is difficult to design molecular-based trials for a number of reasons, he says. For example, some cancers are less common and may take a long time to accrue via the traditional specific histology-based approach.
“Other difficulties include tumor progression and ‘clonal evolution,’ which means that cancer may change over time, especially with treatment,” Dr. Grivas says.
NCI-MATCH attempts to address that hurdle by mandating a new tumor biopsy and genomic sequencing for each patient, which may provide more contemporary and relevant tumor biology information than would archival cancer tissue.
“The standardization and efficiency in several logistical aspects, genomic sequencing platforms, timely bio-informatics data analysis and prioritization algorithms render NCI-MATCH one of the most important and groundbreaking current trials,” Dr. Grivas says. “Other potential benefits include identification of genomic alterations that might qualify patients to future targeted agents not currently available or agents available in another trial.
“We also believe that this study will provide tremendous insight into the molecular underpinnings of various cancer types, thus further advancing the dynamic field of cancer research in the 21st century,” he says.
The trial is funded by the NCI in a unique collaboration with pharmaceutical companies who provided trial agents via agreements with the NCI.
A pediatric version of NCI-MATCH is in development and is expected to launch in 2016.
If you have patients who may be candidates for NCI-MATCH, please contact Vicky Edmisten (216.445.9347 or edmistv@ccf.org), or Amy Pritchard (216.442.5296 or pritcha@ccf.org) for assistance.
NCI-MATCH enrollment information can be accessed online at OnCore (Case Comprehensive Cancer Center’s On-line Clinical Oncology Research Environment) as EAY131-CCF. Registration is required.

First-of-its-kind research investigates the viability of standard screening to reduce the burden of late-stage cancer diagnoses
Study demonstrates ability to reduce patients’ reliance on phlebotomies to stabilize hematocrit levels

Findings highlight an association between obesity and an increased incidence of moderate-severe disease
Cleveland Clinic Cancer Institute takes multi-faceted approach to increasing clinical trial access
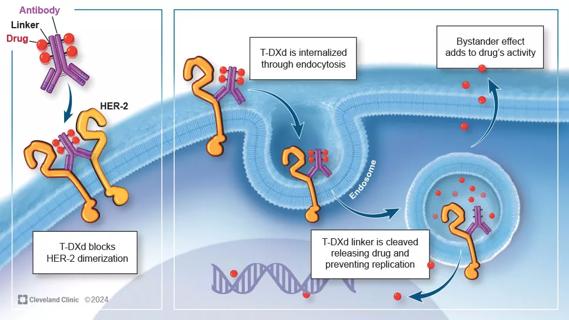
Key learnings from DESTINY trials

Gene editing technology offers promise for treating multiple myeloma and other hematologic malignancies, as well as solid tumors

Study of 401,576 patients reveals differences in cancer burdens as well as overall survival

Enfortumab plus pembrolizumab reduced risk of death by 53% compared with platinum-based chemotherapy