IV infusion associated with lower mortality, shorter ICU stays
More than 1.7 million Americans develop sepsis every year and incidence of the condition is on the rise. In severe cases, widespread inflammation leads to multiorgan failure and death. Results of a new study hold the tantalizing prospect that, for some patients with sepsis, intravenous (IV) vitamin C could increase survival and shorten intensive care unit (ICU) stays.
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
The findings, published in JAMA, are from the CITRIS-ALI (Vitamin C Infusion for Treatment In Sepsis-Induced Acute Lung Injury) trial. Cleveland Clinic was one of seven enrollment sites across the country chosen for the study because of the diversity of its patients and the number of cases of sepsis it sees.
Conducted from September 2014 to November 2017, CITRIS-ALI was the first randomized, double-blind, placebo-controlled multicenter trial to assess vitamin C and its potential impact on sepsis and acute respiratory distress syndrome (ARDS).
“We hypothesized that IV administration of vitamin C would lead to a decrease in inflammation, and hence, improved outcomes,” said co-author Abhijit Duggal, MD, MPH, MSC, Director of Critical Care Research. “But the organ dysfunction scores and inflammatory markers in patients who received vitamin C was not much different from that in the placebo group. However, there were significant differences in the hard outcomes of mortality and length of stay.”
In the study, 167 ICU patients with sepsis and ARDS who had been hospitalized for less than 24 hours were randomized to IV infusion of 50 mg/kg vitamin C in dextrose 5% in water or placebo every 6 hours for 96 hours. The primary outcomes were change in organ failure from baseline to 96 hours and plasma biomarkers of inflammation at 0, 48, 96 and 168 hours.
Infusion of high-dose vitamin C resulted in no significant reduction in change in mean modified Sequential Organ Failure Assessment score from baseline to 96 hours (9.8 to 6.8 intervention vs 10.3 to 6.8 placebo) or in C-reactive protein levels (54.1 vs. 46.1 µg/mL) and thrombomodulin levels at 168 hours (14.5 vs. 13.8 ng/mL), compared with the placebo group. Looking at secondary outcomes, however, the researchers found significant between-group differences favoring vitamin C in three areas: 28-day all-cause mortality, ICU-free days to day 20, and hospital-free days to day 60.
At day 28, mortality was 29.8% in the vitamin C group versus 46.3% in the placebo group (χ2 = 4.84; P = .03); between-group difference, 16.58% [95% CI, 2% to 31.1%]). The number of ICU-free days to day 28 was 10.7 in the vitamin C group versus 7.7 in the placebo group (mean difference, 3.2; 95% CI, 0.3 to 5.9; P = .03). The number of hospital-free days in the vitamin C group versus the placebo group was 22.6 vs 15.5, respectively (mean difference, 6.69; 95% CI, 0.3 to 13.8; P = .04).
“Patients in the control arm were much more likely to die within the first three to five days in the ICU than were the patients on vitamin C. The mortality benefit associated with vitamin C seemed to continue for the duration of the ICU course,” says Dr. Duggal.
While emphasizing that the study was not powered to assess mortality and length of stay, Dr. Duggal points to the Kaplan-Meier survival curve of the two arms as suggestive of the biological plausibility of the impact of vitamin C on sepsis outcomes. “The separation between the intervention and the control arms happened early in the course of the ICU stay, at the time the infusions were occurring. In the graph, both arms stayed pretty parallel for the rest of the patients’ ICU stays.”
Because results for mortality and length of stay are considered exploratory, Dr. Duggal says, they have not changed day-to-day practice at Cleveland Clinic for critically ill patients with sepsis.
“These are intriguing results but physicians should be mindful that we cannot say that vitamin C should be considered standard of care for every patient with sepsis,” says Dr. Duggal. “The idea that something as simple as this could decrease mortality associated with sepsis and ARDS—which is 30% to 35% in most cases—is very tantalizing to us. But additional research is needed to prove that. If it is the case, vitamin C would have the potential to become a front-line drug for patients with these conditions.”
CITRIS-ALI was supported by the National Heart, Lung, and Blood Institute, the National Center for Advancing Translational Science Award, National Institutes of Health, Virginia Commonwealth University (VCU) Investigational Drug Services, and McGuff Pharmaceuticals. In addition to Cleveland Clinic, the other enrolling academic centers were Emory University, VCU, Medical College of Wisconsin, and the University of Kentucky.
Cleveland Clinic researchers are also a part of a multicenter study assessing the role of vitamin C in treatment of patients with sepsis in VICTAS. The VItamin C, Thiamine And Steroids in Sepsis (VICTAS) Study is a double-blind, placebo-controlled, adaptive randomized clinical trial being conducted at 43 academic centers. VICTAS is designed to investigate the efficacy of combination therapy using vitamin C, thiamine and corticosteroids in reducing mortality and improving organ function in critically ill patients with sepsis.

Lessons learned from cohorting patients and standardizing care
New tools and protocols to improve care
Cleveland Clinic pulmonologists share a framework for how to implement effective clinical protocols to standardize evaluation and management of complex acute respiratory distress syndrome

An interdisciplinary team of specialists improves outcomes with effective collaboration, communication and identifying and implementing best practices
A public health tragedy with persistent pathophysiological and therapeutic challenges
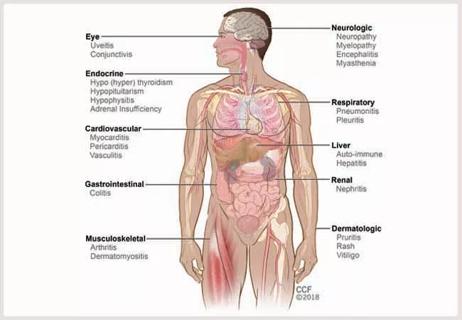
Immune toxicity remains a diagnosis of exclusion, and multidisciplinary collaboration remains the cornerstone for early diagnosis and treatment.

A closer look at risk, clinical manifestations, treatment and prevention efforts
Refer early, even when patients aren’t yet transplant candidates