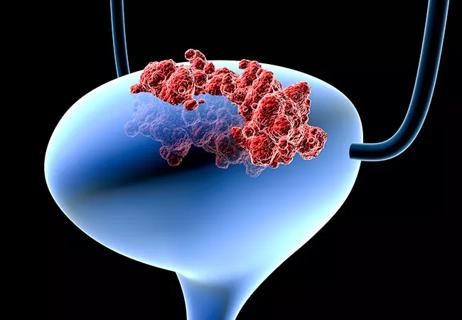
Standard of care is vital as are new therapies
A new standard treatment option for advanced/metastatic cases

Immune and genetic biomarkers predict response rates

Human antibody pembrolizumab offers new hope to patients
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy

Focus is on biology of the cancer, not its location

Cleveland Clinic researchers study new treatment methods