Locations:
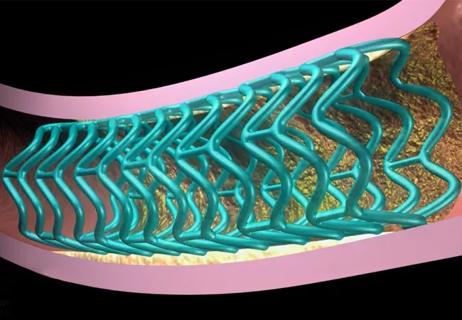
With 5-year results now in, a lead investigator looks ahead
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
Advertisement