A case-based discussion of efficacy, eligibility and use

Editor’s note: This is a reprint of an article originally published in Cleveland Clinic Journal of Medicine. The original, including references, can be found here.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
By Alexa Nicole Fiffick, DO, MBS, NCMP; Tara K. Iyer, MD, NCMP; Tiffany Cochran, MD, MA, NCMP; and Pelin Batur, MD, FACP, NCMP
ABSTRACT
With high rates of unintended pregnancy in the United States, it is crucial for clinicians to be well-informed about the full spectrum of contraceptive options to improve reproductive autonomy. We review new contraceptive options including a nonhormonal intravaginal gel, hormonal contraceptives in the form of new pills, patches, and vaginal rings, and combined hormonal contraceptives that contain new estrogens as alternatives to ethinyl estradiol. We review updated prescribing methods for several established hormonal contraceptives such as depot medroxyprogesterone acetate, which is now available for subcutaneous self-injection. Additional choices of available contraceptive methods have important clinical implications that may remove unnecessary barriers to contraceptive use.
Despite widely available contraceptive methods, the percentage of unintended pregnancies in the United States remains stagnant at 45%, higher than in many industrialized nations, with many of these pregnancies in individuals with subpar access to healthcare.1
Several new contraceptive methods have become available in the last few years, and clinicians should be able to counsel on the full spectrum of options, as patient satisfaction with the method will improve adherence. Particularly now, with reproductive autonomy being discussed in many states, there is a need for action to ensure that every patient receives their preferred safe and effective contraceptive. In addition to preventing pregnancy, contraceptives often are used for symptom management in medical conditions including acne, hirsutism, dysmenorrhea, heavy menstrual bleeding, menstrual migraine, and perimenstrual mood disorders. Herein, we discuss several new hormonal and nonhormonal contraceptive options.
Advertisement
A 47-year-old with latex allergy presents to the office. She is interested in contraception, but does not want anything that contains hormones.
Until recently, nonhormonal contraceptive options were limited to the copper intrauterine device (IUD), condoms (male and female), diaphragm, cervical cap, and spermicides.2–4 The use of a copper IUD requires a procedure and may lead to increased menstrual bleeding and cramping.5,6 Barrier methods are user-dependent, may impact spontaneity, have varying effectiveness, and variable unintended pregnancy rates per year of use (eg, 18% to 28% for spermicide).2–4 Most barrier methods require an office visit for optimal fitting and need to be used with spermicides, which may be associated with irritative vaginal or urinary symptoms.3
A new barrier method intravaginal gel (Phexxi; lactic acid 1.8%/citric acid 1%/potassium bitartrate 0.4%) is inserted within 1 hour prior to intercourse. The gel maintains the physiologically acidic pH of the vagina to inhibit sperm motility, and the viscosity offers a barrier to sperm over the cervix.4 A new applicator needs to be used with each act of intercourse.
The AMPOWER study was a multicenter, single-arm, open-label, phase 3 study of 1,384 women, of which 1,114 were included in the primary efficacy analysis.7 In this study, the intravaginal gel demonstrated 86.3% contraceptive efficacy with typical use (as opposed to preliminary studies submitted to the US Food and Drug Administration [FDA] that suggested 93% efficacy with perfect use over 7 cycles).7–9 When using contraceptive methods with lesser efficacy, an advanced prescription for an emergency contraceptive pill (such as ulipristal acetate) is recommended, especially if pregnancy prevention is an important goal for the patient.
Advertisement
The most common adverse events have been reported to be vaginal burning (18%) and itching (14.5%).9 Similar to other barrier methods, an association with cystitis, pyelonephritis, and urinary tract infections have been reported, perhaps due to the shift in pH of the genitourinary system.9,10
Intravaginal gel is an option for patients desiring nonhormonal birth control (owing to preference or medical contraindication), wanting an on-demand option, or those who have allergies to other barrier methods such as latex condoms or spermicide. This method is particularly unique because it is on-demand and similar to condoms but not partner-dependent. It can also be used postpartum, post-miscarriage, or post-abortion. In addition, some women who are encouraged to use two methods of contraception (ie, when on a medication that will induce liver enzymes or has teratogenic potential) can use this method with a shorter-acting method such as birth control pills. This is a reasonable contraceptive option for women in the late menopause transition, where the chance of unintended pregnancy is decreased (though still possible). Those who need a highly reliable method to prevent pregnancy should be counseled on the use of more effective methods.
A 28-year-old shift-worker notes difficulties remembering her birth control pills. She is interested in an option that does not require daily use. You suggest an IUD or arm implant, but she wants to avoid a procedure. What method would you recommend?
Despite the variety of available contraceptive options, imperfect adherence remains an issue that decreases effectiveness. Nearly 48% of unintended pregnancies occur in women who use contraception.11,12 Most American women use short-acting hormonal contraceptives or combined oral contraceptives (COC), even though the chance of unintended pregnancy is significantly lower for an IUD and implant.5,6 Expanding the available non-oral formulations of short-acting hormonal contraceptives is crucial to ensure that women who do not desire a procedure or daily oral regimen have adequate efficacious options.
Advertisement
A new 13-cycle combined vaginal ring containing 150 μg segesterone acetate and 13 μg ethinyl estradiol provides one year of birth control using the same ring without being discarded (Annovera),13 unlike other vaginal contraceptive rings (eg, NuvaRing, EluRyng) that have a different type of progestin and are intended for only 1 month of use at a time. Similar to other vaginal rings, Annovera is placed inside the vagina for 21 days by the patient, then removed for 7 days to allow menses.13–15
The initial phase 3 trials included two multicenter, multinational studies of 3,052 women, with 999 completing 13 cycles of use with Annovera.16 Effectiveness in pregnancy prevention was noted to be 97.3% when used as recommended, comparable to perfect use of COCs.13,14
Complete expulsions occurred in the studies in 7% of cycles, although these were mainly in the initial cycle.16 No backup contraception is needed if the intravaginal ring is reinserted within 2 hours of expulsion.17 This may not be an appropriate method for those with difficulty or pain using the vaginal ring, such as those with arthritis of the hands, pelvic floor dysfunction, vaginismus, preference to avoid intravaginal placement, or other limitations for correct use.
This method is a good choice for those who want longer-acting, reversible contraception that is not a pill, injection, or procedure, especially in patients struggling to adhere to a daily pill or with limited access to healthcare. The segesterone acetate and ethinyl estradiol ring has not been studied for continuous use (no placebo break), though it is used this way in clinical practice. A modeling study using pharmacokinetics data from 37 women suggested that serum levels of estrogen and progestin after 364 days of hypothetical continuous use would be appropriate for pregnancy prevention,15 but evidence from clinical trials to support continuous use is currently unavailable.
Advertisement
A 35-year-old needs contraception and would like to regulate her cycles. She has a history of lower extremity venous thromboembolism following intensive care unit hospitalization for viral pericarditis. She wants to avoid procedures or injections and prefers oral pills. She has tried a progestin-only pill in the past, does not recall its name, but does remember frequent breakthrough bleeding with that method.
Before 2019, the only progestin-only pill (POP) available in the US would have been 0.35-mg daily norethindrone, available in multiple generic names. Because it does not contain estrogen, a POP can be used in women with a history of arterial or venous thrombosis and those considered to be at high risk for these conditions (ie, uncontrolled hypertension, tobacco use, antiphospholipid syndrome, migraine with aura, etc.).5,6 Given that there are only a few contraindications to its use (such as active hormone-dependent breast cancer treatment), a POP can be offered as a “quick start” method to any woman who is sure that she is not pregnant and as a bridge for more effective methods requiring a procedure at a future time.
With traditional POPs, patients need to follow rigid daily timing as missed pills lessen effectiveness.18 Traditional POPs are different from estrogen-containing contraceptives in a variety of ways. They inhibit ovulation only in about 50% to 70% of user cycles.6,18 Thus, this method also prevents pregnancy with other mechanisms of action, such as impacting cervical mucus.18,19 Norethindrone-only pills also have a shorter half-life than COCs.18 Consequently, a POP user must adhere to a consistent pill schedule, with a maximum delay of 3 hours in dosing compared to 12 to 24 hours with a COC.18,19 Women who are more than 3 hours late taking their norethindrone POP are encouraged to use additional backup contraception.18
The new 4-mg drospirenone POP, a derivative of spironolactone, acts as an antimineralcorticoid with antiandrogenic properties,18,19 with activity similar to 25-mg spironolactone. Therefore, it is likely to be beneficial in those with acne, hirsutism, or tendency toward fluid retention. Although it has not been directly compared to norethindrone POPs, the 4-mg dose for drospirenone was determined based on effective suppression of ovulation for up to 24 hours following a missed or delayed dose.18
Norethindrone POP is taken daily, without any placebo breaks. Thus, variable bleeding can occur, as can lighter regular bleeding, amenorrhea, or irregular spotting. Drospirenone is taken in a 24-active pill/4-placebo-pill formulation to induce a regularly scheduled withdrawal bleed as opposed to traditional POPs taken continuously.19,20
As with all progestin-only methods, unscheduled or prolonged bleeding is common. It typically occurs in the first 6 months, between cycles 2 to 4, with declining frequency over time, and an eventually greater percentage of participants reporting lighter cycles or amenorrhea at a year or beyond.19,21,22 Based on a multicenter, noncomparative trial, the 4-mg drospirenone “24/4-day regimen” reduced unscheduled bleeding over time; participants reported 90% of bleeding days as light or moderate, with only 4.2% of participants stopping the study due to irregular bleeding.19,21,22 Prior to prescribing, appropriate counseling of expected bleeding patterns over the first year is likely to help with patient adherence and satisfaction with the method.
Similar to traditional POPs, drospirenone has a desirable safety profile.19,21,22,23 In a study of over 700 participants from 41 European sites, treated for one year, no one experienced cardiovascular events, thromboses, or hyperkalemia, despite participants having at least one cardiovascular risk factor.19 Drospirenone 3 mg is available in a COC together with ethinyl estradiol doses of 20 or 30 μg (with additional FDA approval to treat premenopausal dysphoric disorder).23 Despite the higher dose of 4-mg drospirenone in the progestin-only formulation, plasma concentrations of drospirenone at a steady state have been shown to be higher after use of 3-mg drospirenone combined with ethinyl estradiol.23 However, this difference is unlikely to have significant clinical implications for most individuals. Given its effectiveness in ovulatory suppression, predictable bleeding pattern, and favorable side effect profile, a 24/4-day regimen of 4-mg drospirenone would be an excellent option for women with contraindications to estrogen-containing hormonal contraceptives.
A frustrated 38-year-old visits the office for a birth control follow-up. She has tried multiple COCs with different progestin types, yet each caused her to develop a diffuse rash. She is adamant about not placing an intravaginal ring, arm implant, or intrauterine device. Are there any other options?
Most COCs contain ethinyl estradiol, a synthetic hormone with a long half-life, which helps with stability of dosing for effectiveness and bleeding control. Oral intake of ethinyl estradiol impacts the production of various liver proteins involved in coagulation, fibrinolysis, and hypertension, thus contributing to its commonly known thrombosis risk. Also, ethinyl estradiol is highly potent compared to more natural analogs of estradiol, which has led to a lowering of ethinyl estradiol doses over the years to improve the safety profile of hormonal contraceptives.24 Although most women tolerate ethinyl estradiol-containing pills very well, for those who are intolerant, there were few alternative estrogen-containing contraceptive options until recently. In 2010, the FDA approved COCs containing estradiol valerate, followed by the recent approval of estetrol in 2021, expanding the number of options for those intolerant to the estrogen component of the pill.
Estradiol valerate is available in a quadriphasic formulation (US trade name Natazia),which decreases estrogen from 3 mg to 1 mg and increases the progestin (dienogest) from 1 mg to 4 mg, both over the course of the monthly regimen. Estradiol valerate is a synthetic prototype of natural 17 beta-estradiol, being rapidly metabolized to estradiol after oral intake.25–27 Two mg of estradiol valerate has the impact of 10 μg of ethinyl estradiol, qualifying this contraceptive as very low-dose.27 Dienogest is a progestin with a 17-cyanomethyl group, causing its strongly progestogenic and weakly antiandrogenic activity.25 Two inert (placebo) pills complete the pack for the last 2 days to allow for a shortened withdrawal bleed.26–28 Comparable to triphasic pills, the 26 active pills contain tapering doses of the active drugs in attempt to mimic the natural menstrual cycle.26–28 Although both estrogen and progestin components are newer to the US market, this particular progestin has been used in Europe since the 1990s.
Natazia was the first quadriphasic dosing regimen used to treat heavy menstrual bleeding.26,27 A multicenter, double-blind, randomized, placebo-controlled phase III study conducted in Europe and the Asia Pacific demonstrated the effectiveness of the estradiol valerate-dienogest combination in significantly reducing menstrual bleeding and improving productivity and daily activities in women’s lives.26 Though most COCs can treat heavy menstrual bleeding, this formulation has higher rates of amenorrhea compared with typical COCs, with amenorrhea occurring in 19% to 24% of women, making it potentially useful for those plagued with persistent breakthrough bleeding using other regimens.26,27 Also, estradiol valerate caused a significantly milder effect on metabolic parameters than ethinyl estradiol.25–29
Estetrol 14.2 mg is a novel estrogen combined with 3-mg drospirenone (US trade name Nextstellis, 2021). Estetrol is marketed as a “natural estrogen” because it is produced by the fetal liver during pregnancy and acts selectively in tissues (impacts alpha receptors), showing mixed estrogen agonist and antagonist activity.30 Clinically, it has been shown to have minimal impact on the synthesis of coagulation factors, hepatic metabolism, triglycerides, and breast stimulation.24,30 Owing to these differences, estetrol cannot be translated into an equivalent dose of ethinyl estradiol that applies to all tissues. However, contraceptive efficacy of estetrol with drospirenone has been shown to be similar to other marketed ethinyl estradiol-estetrol with drospirenone containing COCs. Estetrol has been marketed as more environmentally friendly due to less accumulation of estrogen metabolites in the urine, with the hopes that it will be less likely to pollute water supplies.31 However, further study is needed to assess which of these factors, if any, will be of clinical significance.
Nextstellis is monophasic (all pills have consistent hormone dosing), available with 24 active pills and 4 days of placebo, leading to an optimum withdrawal bleeding pattern (typically, the fewer placebo days, the shorter the expected bleeding).24,30 Studies have suggested lower venous thromboembolism (VTE) incidence compared to traditional COCs,24,30 but there is need for more extensive studies (in real-world settings, in larger groups) to give a more accurate population estimate of VTE incidence. The additional absolute increase in the risk of venous thrombosis with the use of all estrogen-containing contraceptives (when compared to baseline population rates of VTE) is about 1 of 1,000 or less, in the rare category of risk, and well below the thrombosis risks of unintended pregnancy.5,32 Thus, large populations need to be studied to assess thrombosis risk in otherwise healthy women during reproductive age.
The side effect profiles of these newer estrogens are similar to all other classes of combined hormonal contraceptives and may include mood changes, irregular bleeding, nausea, headache, and breast tenderness. Newer estrogen options might be beneficial in those with intolerance to multiple COCs, both in terms of symptoms and bleeding irregularities, and may be the desired option for those with cardiometabolic risk factors such as elevated triglycerides, pending further study. Estradiol valerate, in particular, could be a highly efficacious hormonal contraceptive for women with menorrhagia.
A 23-year-old needs birth control that will provide predictable cyclical bleeding and help her acne, but does not want to be required to remember pills. She has read that birth control patches increase the risks of blood clots more than other formulations and asks for your advice.
The delivery of hormonal contraceptives has evolved since the birth control pill was first introduced in the 1960s, now including oral, transdermal, subcutaneous implant, intrauterine, and intravaginal options. Transdermal delivery of hormones helps address poor adherence and fluctuation of hormones due to pharmacokinetic variations of serum hormonal levels associated with COCs.33–36
Similar to a bandage, a small adhesive patch is placed on the lower abdomen, buttocks, upper arm, or upper torso (excluding the breast), worn continuously for 1 week, and is removed and replaced immediately by a new patch weekly for 3 weeks, followed by a patch-free week when the menstruation occurs. Alternatively, a new patch can be placed weekly without any breaks for continuous use. However, pharmacokinetic studies have suggested a gradual rise in serum ethinyl estradiol over time after 12 weeks of continuous use, thus long-term safety of patch use without placebo breaks is not clear.33–36 The patch is designed to stay in place while bathing, swimming, or exercising, but users should not apply lotion or oil near the patch site. Like all transdermal medications, allergic reactions to the adhesive are possible.
The first patch containing 35 μg ethinyl estradiol and the progestin norelgestromin was FDA approved in 2001 within the United States and internationally (initial trade name Ortho Evra, now Xulane). Once applied to the skin, a steady state is reached within 48 hours of application and maintained consistently until removal; like forms of hormonal contraceptives, the contraceptive effectiveness begins after 1 week of use.6 Several clinical trials indicate that the contraceptive patch is as effective as COCs and may lead to better adherence.34–50 However, this first generation of patches led to higher exposure of serum ethinyl estradiol, similar to a 50-μg COC,36 leading to conflicting reports about whether rates of VTE were increased compared with COCs.37 Some of these studies found no associated increase in VTE risk compared with low-dose comparators.38–40 While another found comparable VTE risks to levonorgestrel-containing pills (a progestin that typically shows lower relative risks of VTE among COCs) but could not exclude additional risk for older women.41 In contrast, several studies suggested that the norelgestromin-ethinyl estradiol patch magnifies relative risk for VTE compared with norelgestromin or levonorgestrel-containing pills by 2-fold.42–44 However, there were limitations to several of these studies, including that the 95% confidence intervals (CIs) crossed one in most analyses and had no adjustment for possible confounding variables.36 Thus, the FDA Advisory Board concluded that though there may be an increased relative risk for VTE with the norelgestromin-ethinyl estradiol patch compared with some birth control pills, the absolute risk is still considered lower.45
A new transdermal patch containing 30-μg ethinyl estradiol and 120-μg levonorgestrel daily dosing (Twirla; FDA-approved 2020) was designed to address the need for a lower-dose contraceptive patch and delivers daily hormone exposure compared with similarly dosed pills.35 Twirla is well tolerated, with reported lower rates of detachment and site irritation than the norelgestromin-ethinyl estradiol patch.34,35 Based on evidence from five clinical trials, the norelgestromin-containing patch has a VTE frequency of 53 per 100,000 women (95% CI 1–294).34 Though direct head-to-head studies are not available between the 2 patches, studies of the levonorgestrel-containing patch are estimated to have a VTE frequency of 32 per 100,000 women (95% CI 1–176).34
Norelgestromin patch users typically had less breakthrough bleeding and spotting than COCs at cycle 13.45 However, users of this patch were also more likely to report breast discomfort, dysmenorrhea, nausea, and vomiting.29,40,41 Users of the novel levonorgestrel patch were less likely to experience nausea and infrequently reported headaches and fatigue when compared to COC users.34,47,48 Again, it should be noted that studies directly comparing side effects of the 2 patches are not available.
Based on limited evidence, there is concern of a higher risk for contraceptive failure in transdermal contraceptive users who have a body mass index (BMI) ≥ 30 kg/m2.35 Thus, relying on a hormonal contraceptive patch as the sole method to prevent pregnancy in women with a BMI ≥ 30 kg/m2 is not recommended, especially when pregnancy prevention is a high priority.
Transdermal hormonal contraceptives are advantageous for individuals having difficulty with remembering a daily pill. Healthy women in their teens and 20s at low risk for VTE may still be candidates for norelgestromin patches, especially if higher doses are preferred for cycle control, improving acne (which tends to be treated best with higher estrogen-containing options), or combined with medications that may interact to decrease hormone levels. However, additional transdermal options with a lower estrogen dose will be useful in clinical practice.
Depot medroxyprogesterone acetate (DMPA) users represented 2% of US women aged 15 to 49 between 2017 and 2019.51 DMPA-intramuscular 150-mg injections have historically required an office visit, which can pose additional barriers to staying consistent with a patient’s preferred contraceptive method. Following their review, the Centers for Disease Control and Prevention and World Health Organization recommended self-administered 104-mg subcutaneous (SC) DMPA.50 This formulation can be injected at home by the patient, which empowers self-care and removes barriers such as frequent in-person visits to a clinic. It is a user-controlled method with the potential to improve contraceptive access and reproductive autonomy.49
Currently, the FDA label states DMPA-SC is only to be administered by clinicians. Nonetheless, following a shared decision between clinician and patient, an FDA-approved drug may be prescribed for off-label use, not excluding self-administration when medically needed. In clinical practice, many patients are encouraged to self-inject at home, and self-administered DMPA-SC has been shown to have a higher continuation rate than provider-administered DMPA.49,51 One study reported that 97% of patients found DMPA-SC easy to administer and reported an 87% satisfaction rate, which was higher than provider-administration at 12 months of follow-up.51 In a US study conducted during the COVID-19 pandemic, 37% of contacted DMPA-intramuscular patients were interested in self-administration of DMPA-SC, with 58% of those interested individuals transitioning to self-administration, reported to be similar to provider administration, and the safety profile was not different, though more injection site reactions have been reported in the self-administered group.52 Barriers to self-administration include a preference for an in-person visit, fear of needles, incorrect administration, and insurance coverage.49
The recommendation for self-administered initiation, follow-up, and reinjection interval is the same as when provider administered. Repeat DMPA injections should be provided every 3 months and may be given up to 2 weeks late without requiring backup contraceptive protection.53 Patients should receive instruction for self-administration and sharps disposal. These individuals should have access to follow-up care along with the opportunity to switch to provider-administration or another contraceptive method if desired.
Long-acting reversible contraceptives (LARCs) have become an increasingly popular contraceptive choice because of their high degree of efficacy and safety profiles that include efficacy at preventing pregnancy, ability to be used for several years, limiting patient effort and thus user error, and offering rapid return to fertility. A prospective cohort study of over 9,000 women with normal weight and BMI > 30 kg/m2 received the LARC of their choice (an IUD or arm implant) and observed a failure rate of less than 1 per 100-woman years without any difference according to BMI category.53–55
When long-term pregnancy prevention is priority, LARC may be the optimal contraceptive choice. Recently, research has shown that several commonly used LARC methods maintain efficacy 1 to 2 years after the FDA-approved duration of use (Table 1).5,6,53,56
Nexplanon is a small polymer rod impregnated with the progestin etonogestrel. During a simple office procedure, this implant is placed subdermally in the non-dominant upper arm. While FDA-approved for only 3 years for contraception, emerging data have shown efficacy maintained for up to 5 years. An open-label, multicenter, randomized trial demonstrated that etonogestrel implants had a 5-year cumulative pregnancy rate of 0.6 per 100 women-years [n = 204; 95% CI, 0.2–1.8].57 In contrast, the chance of unintended pregnancy at the end of 1 year is 6% to 9% with typical use of patient-controlled methods such as pills, rings, patches, and DMPA injections.6 Thus, even when used two years beyond the removal date, it is likely that contraceptive efficacy is considerably better than those of the short-term reversible methods.
In our experience, it is reasonable for a clinician to have a shared-decision-making conversation regarding the option to leave the device for 4 or 5 years or replace it at year 3. Of note, there is insufficient data regarding the efficacy of the etonogestrel implant in extended use in patients with a BMI ≥ 30 kg/m2, as obesity has been shown to decrease serum levels of progestin.5,6 Additionally, given that the progestin dosage decreases over time, some patients who opt for extended use may experience increased irregular bleeding. Before choosing this option, clinicians must discuss with patients to ensure a complete understanding of potential adverse effects.
The highest dose (52 mg) levonorgestrel-releasing IUDs have the longest FDA-approved duration of use of the progestin IUDs available in the United States. Since 2022, both Mirena and Liletta are now FDA-approved for 8 years of use based on updated studies showing effectiveness beyond their originally approved duration of use (had been 5 years). The lower-dose hormonal IUDs (Skyla and Kyleena) should not be used beyond their FDA-approved duration of use due to lack of data.
Studies suggest that the nonhormonal copper IUD (Paragard) remains a highly effective contraceptive at least until 12 years of use.58 In 1997, the World Health Organization and United Nations conducted a large, randomized, multicenter trial that determined the cumulative 12-year intrauterine pregnancy rate was 1.9 per 100 person-years (standard error, 0.6, P < 0.001), which is similar to all sterilization methods.58
Now, more than any other time in recent US history, it is crucial for all clinicians to be well-informed about the full spectrum of contraceptive options. Shared decision-making between each patient and clinician is recommended to choose the best option, recognizing that the risks of any contraceptive are always less than the risks of unintended pregnancy. Though a growing body of evidence supports the safety of expanding the duration of LARC use, individuals should never be coerced into keeping the device longer than what they prefer. The bleeding profiles and contraceptive efficacy may be impacted negatively with a greater duration of use, and for some women, their highest priority may be the best possible protection from unintended pregnancy. Although increasing nonprocedural options with new pills, patches, and rings are important for patient choice, financial barriers to using these newer (and often more expensive) products remain a real challenge. We urge clinicians to advocate on behalf of their patients for various contraceptive options to be made available and affordable to all women who need them.
Advertisement

New guidelines update recommendations

Mode of delivery does not affect patient satisfaction

Approximately 500 million people globally are experiencing 'period poverty'

One approved non-hormonal therapy and another on the horizon reduce vasomotor symptoms
Treatment being offered in cases where medical and hormonal management was not successful
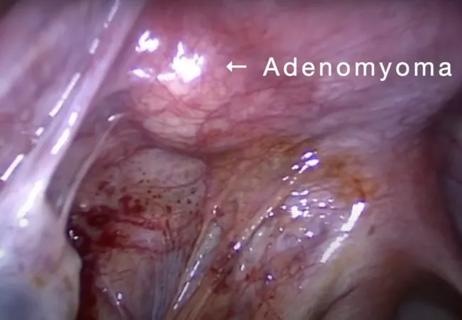
Laparoscopic surgery provides relief for teenage patient with adenomyoma and endometriosis
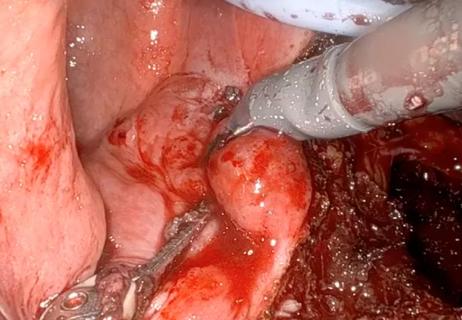
A large nodule had strangulated the patient’s ureter and invaded the vagina, bladder and rectum

Uterine transposition cleared the field for radiation therapy