Correlation found between the biomarker HSD3B1 and resistance to combined hormone therapy and radiotherapy
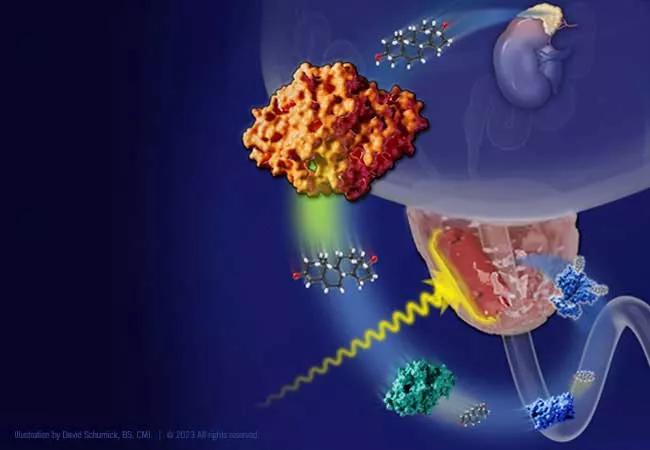
Testosterone biosynthesis pathway
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
Men who inherit a particular type of the steroid biosynthesis enzyme HSD3B1 may exhibit resistance to combined radiation and hormone therapy for prostate cancer, according to a new research study published in the Journal of Clinical investigation.
Historically, there has been a lack of reliable biomarkers to predict response to combined hormone therapy and radiotherapy, a standard treatment for men with high-risk prostate cancer. The enzyme encoded by the gene HSD3B1 is a potential predictive biomarker that may aid clinicians in determining up front whether a patient with prostate cancer has a more aggressive biology that may benefit from intensified hormone therapy combinations, such as adding an androgen receptor selective inhibitor (ARSI) to conventional androgen deprivation therapy (ADT) and radiation.
Nima Sharifi, MD and a team of researchers had initially described germline variants in the HSD3B1 gene, one of which led to more rapid resistance to androgen-deprivation therapy. Building on this work, Dr. Mian and his team studied whether or not this HSD3B1-mediated partial or complete resistance to hormone therapy promotes radiotherapy resistance as well in men with localized, potentially curable disease.
“The stability of the enzyme can dramatically influence the rate at which tumor cells produce testosterone from circulating adrenal precursors, which is critically important for a disease like prostate cancer that is driven by testosterone,” explains study co-author Omar Mian, MD, PhD, a radiation oncologist and physician/scientist at Cleveland Clinic. “The adrenally permissive allele of HSD3B1 can increase the production of testosterone in tumors, bypassing the most commonly used class of testosterone-lowering medications, with the downstream effect of promoting resistance to combined hormone therapy and radiation”.
Advertisement
This pre-clinical and mechanistic study sought to evaluate whether levels of 3bHSD1 (the enzyme encoded by HSD3B1) were associated with radiation resistance. In addition, the study team sought to determine the precise mechanism for this resistance. To do so, the researchers evaluated preclinical models of prostate cancer and specifically interrogated their level of radiation response.
They also explored the influence of 3bHSD1 levels and HSD3B1 genotype on DNA damage repair capacity of prostate cancer cells. They examined the association between androgen signaling and DNA repair pathways in 681 patient samples treated for prostate cancer at Cleveland Clinic for whom there was gene expression data available. Finally, they attempted to reverse the 3bHSD1 mediate radioresistance with targeted hormone therapy combinations.
“There was a clear association between androgen receptor signaling and DNA damage response across the preclinical models as well as patient samples,” says Dr. Mian. “Even low-level testosterone production in the tumor itself, and presumably the region around the tumor, appeared to drive a robust resistance to radiotherapy. This resistance was associated with an enhanced DNA damage repair capacity in cells expressing the adrenally permissive subtype of 3bHSD1.” Importantly, the study team found that this resistance was reversible by treatment with direct androgen receptor targeting therapy with an ARSI.
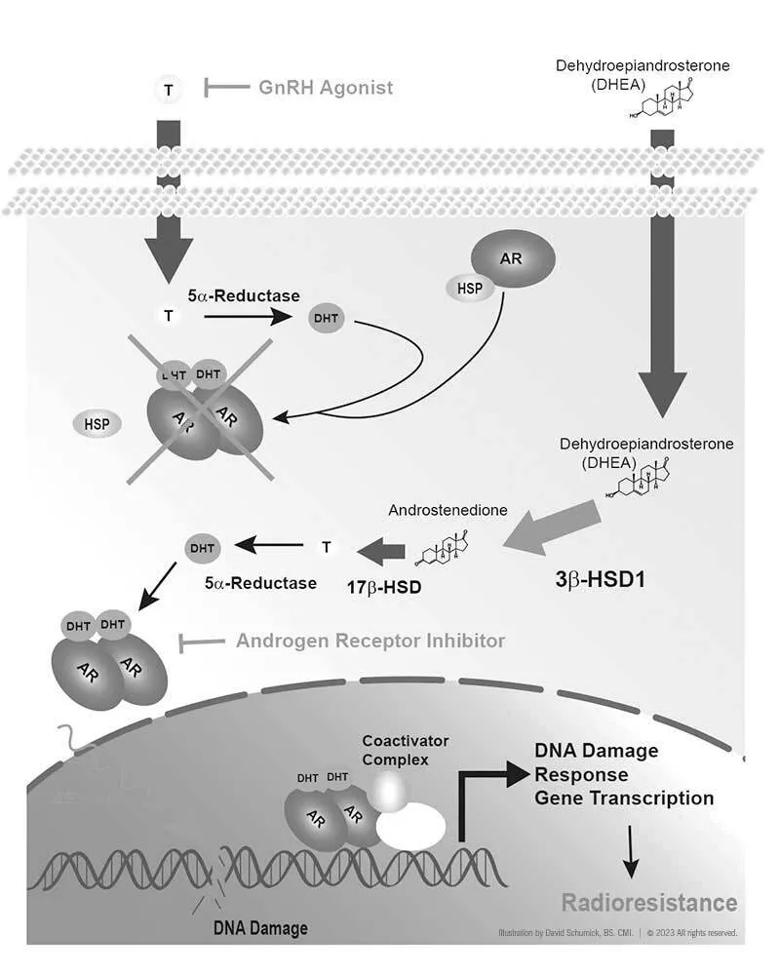
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/0ccb56be-9b1a-464f-89ec-893396faa814/22-CNR-3127708-CQD-Inset2-GREY-800x1000-credits_jpg)
A schematic overview depicting a bypass pathway that renders gonadal androgen blockade (GnRH agonist/antagonist) ineffective via upregulation of HSD3B1 in prostate cancer cells. This in turn promotes resistance to radiation therapy. The bypass pathway is targetable through the use of androgen receptor inhibitors, which also restore sensitivity to radiation.
Advertisement
The researchers are now conducting a prospective registry of the HSD3B1 genotype to validate the study findings in patients receiving combined radiation and hormone therapy. The center is enrolling patients with high-risk, localized disease and low-volume metastatic disease, for whom radiotherapy to the prostate combined with androgen-deprivation therapy is a standard of care. The main clinical implication of this work is that a germline biomarker may identify a high-risk subset of men who would benefit from targeted therapies such as enzalutamide, apalutamide and bicalutamide combined with conventional hormone therapy and radiation.
These studies may lead to routine use of genotyping of enzymes like HSD3B1 to risk stratify patients and match them to the appropriate treatment. This could result in intensifying treatment for patients with high-risk disease, or scaling back treatment for patients who harbor a less aggressive genetically-defined variant of prostate cancer, to spare them the side effects of more intense treatment.
“With a disease like prostate cancer that has a wide spectrum of aggressiveness, having biomarkers that can help us to more precisely match patients to the most appropriate treatment is important,” says Dr. Mian.
Advertisement
Advertisement

Combining advanced imaging with targeted therapy in prostate cancer and neuroendocrine tumors

Approach offers a ‘middle ground’ between radical prostatectomy and active surveillance

AI histologic classifier reliably predicts clinical risk in men post-prostatectomy

Historic collaboration connects two Cleveland Clinic locations, enables real-time sharing of metrics and surgical progress
Patient factors and cancer characteristics are key to deciding between focal therapies and whole gland treatment

Novel research to evaluate noninvasive treatments in ED and chronic pelvic pain
FDA-approved therapy offers promise for patients with castrate-resistant prostate cancer

Using novel robotic approaches for a difficult-to-access prostate