A mindset shift has changed the way pulmonologists both treat and define PFF

Much has changed over the past few years when it comes to fibrotic lung diseases. As more data has emerged, the thinking around these diseases, how they are defined and diagnosed, has also evolved. Previously, patients with fibrosing lung disease were classified along a spectrum of lung disorders based on the cause of the underlying condition. Now, with the broad application of nintedanib, rather than focusing on the individual disease type, clinicians can focus on the treatment of fibrosis and the initial cause of the scarring. However, while the general approach to disease management has become more uniform, it’s important to still recognize that patients with progressive pulmonary fibrosis present differently and still require individualized treatment plans, and the clinician’s role in identifying when pulmonary fibrosis becomes progressive is still a critical aspect of care.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
“People have started to lump these different diagnoses together into one umbrella term of ‘progressive fibrotic lung disease,’” explains Leslie Tolle, MD, a staff physician in Cleveland Clinic’s Respiratory Institute. “The implication here is that it doesn’t necessarily matter what the specific diagnosis is, but if there is scarring on the lung tissue and it progresses, clinicians can add similar treatments to manage these conditions. This is a fairly recent shift in how we as pulmonologists approach treatment, and this shift stems in part from the INBUILD trial from a few years back.”
The INBUILD trial was a double-blind, placebo-controlled phase 3 trial conducted in 15 countries. A total of 663 patients with fibrosing lung disease that affected more than 10% of lung volume on high-resolution computed topography (CT) scans were randomly assigned to receive nintedanib at a dose of 150 mg twice daily or a placebo. The researchers found that the cohort given nintedanib experienced a rate of decline in forced vital capacity (FVC) at a significantly slower rate than the placebo cohort (-80.8 ml/year versus -187.8 ml/year).
One of the key points of the study was that it did not group patients by individual disease, but rather by the basis of the presence of pulmonary fibrosis on chest imaging and common progressive clinical features. This is similar to the approach clinicians have begun taking of looking not at the cause of scarring, but rather simply the existence of scarring. In addition to the findings about the effectiveness of nintedanib, the trial also introduced standards for what constituted pulmonary fibrosis as progressive.
Advertisement
As part of the trial design, the patients were required to meet at least one of the three criteria for progression of interstitial lung disease within 24 months before screening. This was despite standard treatment with an agent other than nintedanib or pirfenidone. The criteria included:
At the time of enrollment, patients were also required to have an FVC of at least 45% of the predicted value and a diffusing capacity of the lung for carbon monoxide (corrected for hemoglobin) of 30 to less than 80% of the predicted value.
“There still isn’t consensus on what constitutes progression,” explains Dr. Tolle. “However, this trial used a definition for progression that is now fairly common, though still not universally accepted. For the most part, we’ve been following these rules at Cleveland Clinic within reason. With the results from this trial and the way patients with lung scarring — no matter the cause of the scarring— responded to the drug, we’ve incorporated nintedanib into the care path when applicable. The drug has a fair amount of side effects, so we can’t universally adopt it, but we do use it.”
Part of the reason that PFF is challenging to treat is that scarring can be caused by many different factors. These include connective tissue diseases, certain medicines and environmental pollutants and often unknown (also known as idiopathic) causes, among others.
Advertisement
“You can get scarring in your lungs for a lot of different reasons,” says Dr. Tolle. “For example, bird feathers and dust can cause lung diseases like this, and so, part of the treatment recommendations for something like this would be to avoid birds, and sometimes we use prednisone or other immunosuppressives. If they get scarring on top of that, then we can add nintedanib because now it falls under this dogma of progressive-scarring lung disease. But, if you have a disease like asbestosis where there is no underlying treatment — once you’ve inhaled the asbestos particles, there’s no way to expel them — then the only treatment is adding this new medication to it.”
Patients with PFF who come to Cleveland Clinic are treated holistically with a multidisciplinary approach. Pulmonologists often work with clinicians from several other specialties, including pathologists, radiologists and rheumatologists among others, to develop an individualized care path. “Imaging is a major part of the diagnosis process, so radiologists help read the scans, and pathologists will help us read biopsy results and determine the initial diagnosis,” says Dr. Tolle. “Often these patients have other conditions on top of lung scarring. So, for example, some of these patients also have rheumatoid arthritis, so we’ll work in tandem with their rheumatologists to come up with a treatment plan that helps both of these conditions without making one or the other worse. Ultimately, it’s just important to recognize that this disease and patients affected by it are affected differently, so we still need to consider patients and their care on an individual basis.”
Advertisement
Advertisement

A review of treatment options for patients who may not qualify for surgery

Rising rates in young miners illustrate the need for consistent prevention messaging from employers and clinicians

Multidisciplinary focus on an often underdiagnosed and ineffectively treated pulmonary disease

Management and diagnostic insights from an infectious disease specialist and a pulmonary specialist

Treatments can be effective, but timely diagnosis is key
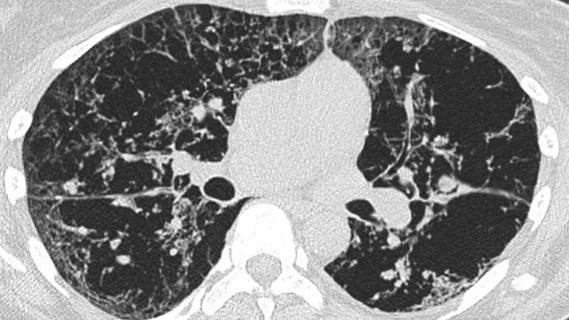
A Cleveland Clinic pulmonologist highlights several factors to be aware of when treating patients
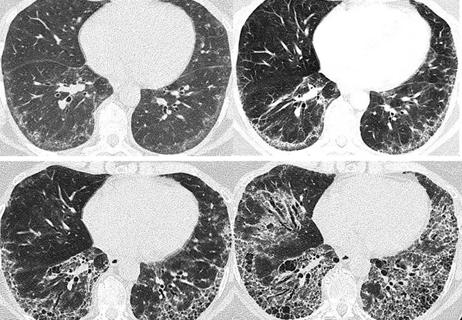
Patient experience improves with a multidisciplinary approach

Collaborative patient care, advanced imaging techniques support safer immunotherapy management