Early diagnosis and referral to a multidisciplinary team are essential
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/a79d933b-2210-4715-a179-17200e84a62c/22-ORI-3296341-CQD-650x450-1_jpg)
Woman Receiving Radiation Therapy Treatments for Breast Cancer
By Amir Hossain Gahanbani Ardakani, BSc, MBBS; Martina Faimali, MBBS, FRCS (Tr&Orth); Lukas Nystrom, MD; Nathan Mesko, MD; Muntzer Mughal, MBChB, ChM, FRCS; Howard Ware, MBBS, FRCS (Tr&Orth); and Panagiotis Gikas, BSc, MBBS Hons, MD (Res), PhD, FRCS (Tr&Orth)
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
This article is reprinted from the July 2022 issue of Cleveland Clinic Journal of Medicine (2022 Jul 1;89(7):393-399).
Treatment of metastatic bone disease has evolved over the last 50 years, but a lack of awareness and recognition of symptoms continues to delay referral to specialist teams. This article highlights crucial concepts surrounding the management of patients with bone metastasis, reviews changes in therapy that have occurred over time, and clarifies the need for a multidisciplinary approach to management. It provides guidance in achieving early diagnosis and referral for patients who present with metastatic bone disease in primary care settings and offers a decision tree for assessing surgical and nonsurgical treatment options.
The estimated cost directly attributed to care of bone metastasis in the United States is greater than $12.6 billion annually, which accounts for 17% of total cancer care.1 In 2012, Medicare paid $100 million in hospital charges to cover the cost of prophylactic internal fixation of the femur as a result of metastasis.2
The rate of new cases of cancer in the United States currently stands at 442.4 per 100,000 men and women per year. In January 2019, the U.S. National Cancer Institute predicted 16.9 million cancer survivors in the United States with a projected increase to 22.2 million by 2030.3
Similarly, in the United Kingdom, the number of people living with cancer is rising by 3% each year, with survivorship projected to increase by 1 million per decade from 2010 to 2040.4 In 2015, an estimated 2.5 million people were living with cancer in the United Kingdom, with a predicted rise to 4 million by 2030.1 In the United Kingdom, 375,000 new cancer cases are diagnosed every year, or about 1,000 new cases daily.5
Advertisement
In the early 1970s, the median survival time for patients with metastatic disease was one year. By 2007 it was six years, and by 2011 it was 10 years.6,7 Today, it is estimated that over 50% of patients survive their disease beyond 10 years.7 With this increased longevity, the age of patients with metastatic bone disease and rates of survival are on the rise.8,9
Incidence of bone metastasis
Bone is the third most common organ affected by metastatic cancer, after the lung and liver.10 Although it is difficult to fully appreciate the incidence of metastatic bone disease, it is estimated that more than 50% of all cancers develop bone metastases, with the variability in the literature ranging from 12% to 70%. In 2008, the incidence of metastatic bone disease in the United States was approximately 280,000 patients per year with an upper estimate of 322,000. This is likely to have increased significantly since then.11
Although almost any carcinoma can metastasize to bone, those that do so most frequently are prostate, breast, renal, lung, thyroid and blood (multiple myeloma) in origin.12 Some autopsy studies have demonstrated skeletal metastases in 90% of men who die of prostate cancer.13 Bone is the most common site of metastasis in patients with breast cancer, and up to 70% of women with metastatic breast cancer have some form of skeletal involvement.14
Quality of life
The quality of life in patients with skeletal metastases is compromised by skeletal-related events (i.e., intractable pain, forced immobilization, hypercalcemia, spinal cord compression, pathologic fractures). Bony metastasis is often the most symptomatic and disabling manifestation of secondary cancer.12 Approximately 68% of patients with skeletal metastasis have pain, and 10% to 20% of those with long-bone metastases eventually sustain pathologic fractures.15,16
Advertisement
Pathologic fracture may be the first sign of disease and the index finding leading to the diagnosis of cancer. In 3% to 4% of patients who present with pathologic fracture, the primary site is not discovered.17 In most cancer types, the morbidity rate in patients with multiple skeletal-related events is higher than in patients with single events. Additionally, the presence of extraosseous disease in the context of skeletal-related events is a powerful predictor of poor outcomes.18
Bone metastasis typically occurs via hematogenous spread and therefore tends to seed in more heavily vascularized parts of the skeleton.15 The most frequent sites for metastases are the spine, pelvis, proximal femur, proximal humerus, skull and ribs,19 and involvement of any of these sites can significantly affect activities of daily living, quality of life, functional status and overall prognosis.15
Healthcare costs
The management of patients with skeletal events due to bone metastasis has important implications for healthcare costs. Early intervention for patients with metastatic bone disease has been shown to reduce patient morbidity as well as overall cost.20 A prompt, proactive response has been shown to reduce complication rates, length of stay, need for community care and overall treatment costs, and this is specifically true of pathologic fracture.20 A prophylactic approach has shown to be safer and much more cost-effective compared with traditional management, or acute fixation, after a completed fracture.21
Advertisement
Patients are living longer with advances in systemic therapy, targeted therapy and radiotherapy treatments, thus making durable reconstruction of a metastatic skeletal location more important. The appropriate surgical approach and choice of implant have the potential to reduce healthcare costs.12
Patients with metastatic bone disease commonly present to orthopaedic surgeons in one of three ways:
Unfortunately, most patients have already sustained a pathologic fracture by the time they present to an orthopaedic team21 and thus have a greater likelihood of severe discomfort and increased morbidity during the treatment process. A fracture event may create complexity that limits treatment options. Heightened awareness in the primary care setting of possible metastatic bone disease is essential in patients who present with musculoskeletal pain and a history of cancer or previous radiotherapy.
Bone cancer pain
The primary symptom often described by patients is pain, and this is especially relevant in a patient with cancer.10
Bone cancer pain can be very complex and has an associated intricate pathogenesis.22 It is often described as a dull ache that is deep and intense in nature, exacerbated by weight-bearing, and often worse at night. Red flags for bone metastasis include a chronic dull ache that continues to worsen over time, pain associated with weight-bearing, night pain, pain on direct palpation, and unexplained localized pain in a patient over age 45. A sudden change to more intense or severe pain usually indicates a pathologic fracture, particularly in the context of minimal trauma.
Advertisement
Patients who live with cancer ultimately deal with considerable suffering and pain; therefore, sudden changes in the quality or quantity of pain should be acted on swiftly.12,22 Significant symptoms that accompany pain include unexplained weight loss, night sweats, and any red-flag symptoms of back pain (e.g., nighttime pain during movement; band-like bilateral nerve root pain or radiculopathy; unsteady gait; progressive weakness of limbs; bowel and bladder symptoms).
Time to metastasis
Few epidemiologic studies establish the median time from primary cancer diagnosis to bone metastasis. The results vary by country, ethnicity, primary cancer type, patient age and initial treatment received.
In broad terms, the highest risk of metastatic bone disease is within the first three to five years of the initial diagnosis, before the cancer reaches a stable state (i.e., no increase or decrease in severity or extent).9,23 However, bony metastasis can present as late as 20 years after the primary diagnosis, so a history of cancer at any stage is important.24
Progression to fracture
Patients with known cancer involving the skeleton or those who have had previous radiotherapy to skeletal metastatic deposits are at particular risk of pathologic fracture. Several landmark studies have suggested that the risk of pathologic fracture after radiotherapy can range from 13% to 41%.25,26 One study suggested that after radiotherapy, 26% of patients develop disease progression at the bony site,27 and another study noted that 35% of fractures develop at just six months after radiotherapy.28 For this reason, patients who receive radiotherapy for bony metastasis should be assessed by an appropriate specialist to determine the need for further stabilization or surgical treatment.
Evaluation
Patients who present in a primary care or hospital setting with suspected metastatic bone disease need preliminary investigations in addition to an initial thorough examination. Certain blood tests (e.g., alkaline phosphatase, lactate dehydrogenase, calcium, blood cell count, basic tumor markers) and plain radiographs can supplement the history and physical examination.12 Although no blood test is specific for bone metastasis, increased calcium and alkaline phosphatase levels can supplement the clinical picture of metastatic bone disease.
Because bone lesions may not become apparent on radiography until 50% to 70% of the bone has been destroyed, initial radiographs may not show an obvious abnormality. A patient with bony lesions may experience symptoms related to hypercalcemia — such as nausea, vomiting, polyuria, muscle weakness, constipation and confusion — and metastasis to the spine may cause neurologic compromise. Urgent referral to an oncologist, orthopaedic surgeon or neurosurgeon is warranted if cauda equina syndrome is suspected.29 Delays in appropriate treatment can lead to increased morbidity, complications and challenges that would not have been present earlier in the disease process, such as changes in bony anatomy with wider destruction, increased frailty of the patient and missed opportunity for less-invasive treatment options.
When bone metastasis is suspected or confirmed, the next step is to establish the origin and nature of the lesion, the degree of disease dissemination, the patient’s overall health and prognosis, and the effect of the lesion on the bone.30 Analysis of this information requires a multidisciplinary effort to allow for effective decision-making as to the most appropriate management (Figure 1).
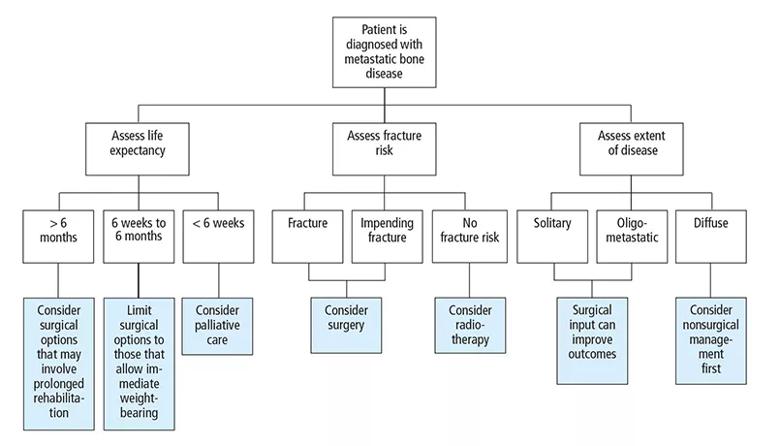
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/715f680e-c1d4-49bb-b5d5-b4ea8e9de650/22-ORI-3296341-CQD-Inset_jpg)
Figure 1. An overview of team management of metastatic bone disease.
The multidisciplinary team
Management of metastatic bone disease requires input from a team of specialists to determine the best treatment options for the individual patient. The team should consist of a medical oncologist, radiation oncologist, radiologist, pathologist, orthopaedic surgeon with an interest in bone metastasis, anesthesiologist, palliative care specialist, rehabilitation specialist, cancer nurse specialist and, most importantly, the patient and family.15
Origin and nature of the bone lesion
The origin and nature of a bone lesion play a key role in the decision-making process. All bony lesions are treated as primary bone tumors, or sarcoma, until proven otherwise. Applying this principle ensures that no primary bone tumor receives delayed or inappropriate treatment.30
Initial investigation: Imaging and biopsy
The initial investigation includes a computed tomography (chest, abdomen, pelvis), whole-body nuclear bone scan, positron emission tomography, and magnetic resonance imaging of the affected area.
Biopsy is becoming a more important and better recognized diagnostic step. Today, most patients who present with a bone lesion should be considered for biopsy in order to obtain a histologic diagnosis, regardless of whether metastatic bone disease is suspected. Biopsy has been reported to reveal a benign diagnosis, infection, a different primary cancer, or change of immunophenotype between the primary disease and the metastasis.12
Bone scan, positron emission tomography and computed tomography are increasing in use, are readily available, and can determine the degree of disease dissemination. This is important because metastatic bone disease can range from a solitary lesion to widespread bone involvement.31
Prognosis
Estimating a patient’s life expectancy and overall prognosis will significantly frame the support and input the patient requires. As a general rule, a patient should have a life expectancy greater than six weeks if surgical management is to be considered. With this prognosis, the surgical procedure must permit immediate weight-bearing. If the procedure requires partial weight-bearing or no weight-bearing postoperatively, the minimum prognosis must be at least three to six months. A life expectancy greater than six months justifies and requires comprehensive surgery (Figure 1).12,29
The key principles of management of metastatic bone disease are to control pain, maintain or improve quality of life, allow early mobilization, create a durable orthopaedic construct to replace or augment bone, and prevent disease progression if possible.32
Painless, smaller lesions
It is generally accepted that painless, smaller lesions with little risk of fracture respond well to radiotherapy alone, but a pathologic fracture will likely require some form of surgical stabilization. A delicate balance is required to avoid overtreatment and undertreatment of these lesions, especially with procedures that have longer recovery times or incur greater morbidity. The clear benefit of operating on early impending fractures must be weighed against the risks of surgery, anticipated prognosis and overall benefit to the patient.33
The primary aim of treating asymptomatic small lesions is disease control and prevention of skeletal-related events. The mainstay of treatment is systemic control such as hormonal therapy, immunotherapy or targeted therapies, chemotherapy, or agents that improve bone strength combined with potential radiotherapy for local control. For smaller symptomatic lesions and those at more difficult surgical locations, percutaneous ablation techniques with interventional radiology have been shown to be effective.34
Larger, symptomatic lesions
For lesions that are larger and more symptomatic, the aim of treatment is not only to control disease but also to maintain mobility and improve pain.32 The need for surgical intervention must be considered in addition to local radiotherapy and systemic medical control of the disease.
Although postoperative radiotherapy has played a role in management, evidence supporting its use is weak, and the associated risks are quite high (e.g., wound infection, skin irritation, osteoporosis and failure of metalwork). Because radiotherapy itself is a risk factor for propagating pathologic fractures, its use needs to be weighed against the potential benefits.35
Antiresorptive drugs are the mainstay of nonsurgical treatment of bone metastasis, and bisphosphonates and denosumab are the most commonly used.
Bisphosphonates and denosumab
Bisphosphonates affect osteoclast activity and survival.36 Zoledronate is approved for use in solid tumors and multiple myeloma, and pamidronate is approved for bone metastases from breast cancer and multiple myeloma. Ibandronate is effective in breast cancer patients. Zoledronate is particularly useful in hypercalcemia associated with bony metastasis. Monitoring is required for complications such as kidney failure, hypocalcemia and osteonecrosis of the jaw.
Denosumab reduces osteoclast activity and is generally well tolerated. It can be used in patients with renal failure since it is not nephrotoxic. It has been shown to prolong the time to first skeletal-related event in patients with metastatic breast and prostate cancer.37
Radiotherapy
Radiotherapy is used primarily for pain management, spinal cord compression and pathologic fractures. Pain relief is achieved within the first two weeks and is almost complete in 50% of patients. The dose, technique and schedule depend on several factors. Short courses of treatment are often used in Europe and Canada, while longer courses are preferred in the United States.37
Other methods
Other methods of pain relief should follow the World Health Organization analgesic ladder38 and range from anti-inflammatory drugs to opiate-based treatment. Guidelines for more detailed pain management options in cancer patients have been published39 and may require input from specialized pain services.
When surgical intervention is necessary, the intervention should be a single procedure that will last the patient’s life span while allowing immediate weight-bearing and mobility.17 Pathologic fractures caused by metastatic bone disease will not heal, even with radiotherapy.
The surgical intervention must be appropriate for the stage of disease, condition of the patient, and the patient’s preferences and wishes. In general, surgical options include the use of intramedullary nails, ridged plate and screws, bone cement supplementation and endoprostheses, or a combination of these.11,17,31
Current research favors early diagnosis and a prophylactic surgical approach in managing bony metastases in patients with impending pathologic fractures. Many studies have shown that in appropriate patients, a prophylactic procedure (compared to a procedure performed after fracture) leads to reduced blood loss, reduced length of hospital stay, quicker return to baseline mobility and, overall, a better two-year survival rate.40
The surgical approaches have evolved with advances in technology and prosthesis design. Fixation alone may not necessarily be the most appropriate option. For example, there is a popular notion that surgical management involves only prophylactic intramedullary nail stabilization. But more recent studies have shown that in appropriate patients, the use of massive endoprostheses for the treatment of bone metastases is a reliable method of limb reconstruction.41 This option is associated with low complication and failure rates, can restore good function, allows for early weight-bearing, alleviates pain and sometimes allows for complete resection of the tumor.41
Observational studies have shown sustained improvement in pain relief and function up to one year after surgery in patients with metastatic bone disease, irrespective of prognosis.42 Studies have also indicated that patients with low-volume bony oligometastatic disease (less than five metastases throughout the body) have enhanced survival and better disease prognosis with appropriate surgical intervention.12,43,44 While these arguments show that the burden of disease and morbidity should not be underestimated, there still exists little awareness and appreciation in hospital and primary care settings regarding possible management options for skeletal-related events due to bone metastasis.12
For a list of references and disclosures, see the original article in Cleveland Clinic Journal of Medicine (2022 Jul 1;89(7):393-399).
At Cleveland Clinic, Dr. Nystrom is staff in the Department of Orthopaedic Surgery, and Dr. Mesko is Center Director, Orthopaedic Oncology, and co-Director of Sarcoma Care. At Cleveland Clinic London, Mr. Ardakani is a research fellow in the Department of Orthopaedic Surgery, Prof. Mughal is Director of the Department of Surgical Oncology, Prof. Ware is Director of the Department of Orthopaedic Surgery, and Mr. Gikas is staff in the Department of Orthopaedic Surgery. Ms. Faimali is from the Department of Orthopaedic Oncology at the Royal National Orthopaedic Hospital in Stanmore, U.K.
Advertisement
The many factors that influence 25(OH)D levels add complexity to questions about hypovitaminosis D
Case study of radial-to-axillary nerve transfer for tumor-related deltoid nerve injury
Study also finds that 26% of children with cancer have mutations in DNA repair genes
Rare genetic disorder prevents bone mineralization
Research highlights promising outcomes for treating recurrent and metastatic cases
Biologic approaches, growing implants and more
Multicenter effort aims to standardize information about diagnosis and early management
Iliac crest structural autograft repairs large, uncontained defect