The importance of anti-vaping awareness

Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/cb57e23d-e73b-40aa-b44e-5575ebe827bf/vaping-e-cigarette-1553043642)
Teen vaping
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
The use of e-cigarettes or vaping devices increased rapidly since their introduction to the U.S. market in 2007. E-cigarettes are battery-operated devices that heat a liquid, generating an aerosol for inhalation. The industry has dramatically expanded the types of e-cigarette devices and the e-liquid formulations to add several flavors that appeal to the youth market, such as fruits, crèmes, mint, chocolate, vanilla, coffee and cookies. In addition, there has been a rise in the vaping of non-nicotine substances, such as tetrahydrocannabinol (THC) and cannabidiol (CBD). Research conducted in 2018 found that young adults were more likely to use flavored tobacco products than adults. In 2019, one in four high school students and 10% of middle school students reported current use of electronic cigarettes—an estimated 4.1 million high school students and 1.2 million middle school students. 1
As of February 18, 2020, the U.S. Centers for Disease Control and Prevention (CDC) reported a total of 2,807 cases of e-cigarette or vaping product use-associated lung injury (EVALI), 15% of whom were < 18 years of age.2 The CDC defines EVALI as the onset of respiratory symptoms within 90 days of using e-cigarettes along with radiographic findings of bilateral ground-glass opacities.
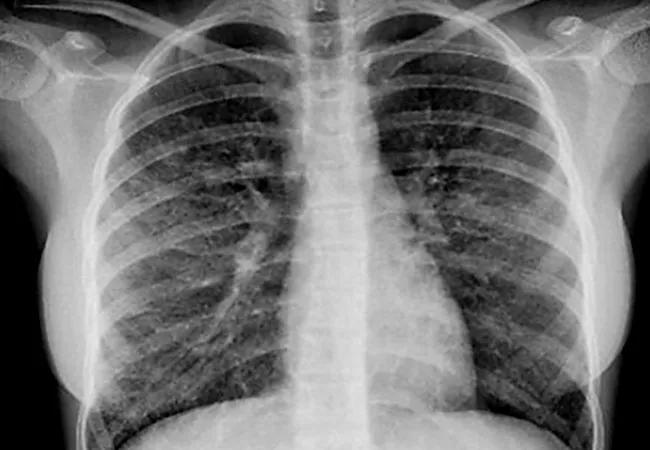
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/6f7c1955-cc28-4712-b244-2df7e49810e0/650x450-Adolescent-Vaping_jpg)
The purpose of our study was to describe our institute’s experience with EVALI.3 We reported a case series of the presenting symptoms, examination findings, results of infectious studies, radiological images and the outcome of 7 cases of EVALI in three female and four male adolescent patients aged 15-18 who presented to Cleveland Clinic Children’s from August-November 2019.
Advertisement
All seven patients reported using e-cigarettes within 30 days prior to hospitalization. Six patients had a history of using refillable e-cigarettes containing THC. One had a history of using disposable e-cigarettes filled with nicotine. The consecutive daily use of vaping products among this group ranged from a period of three months to four years. Patients presented with cough, hypoxemia, respiratory distress, and gastrointestinal and constitutional symptoms. Five of our patients required supplemental oxygen, with four requiring ICU care for additional respiratory support. Three patients were treated with systemic steroids while inpatient. Bilateral opacities were seen on radiographic imaging of all of our patients. The median length of stay was 6.5 days (1-10 days), the median duration of ICU admission was 5 days (3-7 days), and all patients were discharged home on room air. For those who had a follow-up with pediatric pulmonologist, the pulmonary function test showed impaired diffusing capacity of the lungs for carbon monoxide (DLCO). This suggests that although recovery from the acute illness process of EVALI was achieved, there could be long-term impact on lung function associated with vaping.
The adolescent patient population is especially vulnerable to EVALI as vaping becomes more popular and easily accessible among today’s youth. Our patient population had a high prevalence of ADHD, anxiety and depression as co-morbid conditions. These patients may represent a group within the adolescent patient population for which EVALI is potentially higher risk. Further studies are required to investigate this apparent common thread and define the best tactics for reducing substance abuse and vaping.
Advertisement
The presentation of our patients mirrored those reported in nationwide. Both respiratory failure and an exaggerated inflammatory response seem to be the key findings of EVALI. Little is known about the exact pathophysiology of this condition. A study showed that of 51 bronchoalveolar lavage (BAL) fluid samples from patients with EVALI, 94% tested positive for Vitamin E acetate, compared with none of the BAL samples from 99 healthy controls. Intriguingly, a murine model of inhaled vitamin E acetate also showed evidence of lung injury, such as lung epithelial damage.
Parents should be educated on different types of vaping devices. The most popular has been designed to look like a USB flash drive and is rechargeable via a USB port. This design enables users to use their e-cigarette covertly.
Pediatricians must screen and educate patients around the dangers of vaping, including the risk of EVALI during all office visits with adolescents.
Physicians must have a high index of suspicion and keep EVALI in their list of differential diagnoses when evaluating any unwell adolescent patient presenting to the office or emergency room. The initial presenting symptoms of EVALI may not be merely respiratory, as seen in majority of our patients who presented with gastrointestinal symptoms.
Along with our studies, our case series highlights the importance of close follow-up following EVALI. The long-term follow-up of EVALI patients is not well documented owing to the recent nature of EVALI itself. We highly recommend follow-up with a pediatric pulmonologist, including spirometry and DLCO following discharge, to closely monitor this relatively new entity’s recovery process.
Advertisement
Future studies, specifically longitudinal observations, are needed to determine the extent of the clinical outcome and effect on pulmonary function.
Dr. Rezaee is Associate Professor of Pediatrics at Cleveland Clinic Lerner College of Medicine of Case Western Reserve University.
Advertisement
Advertisement
Life-changing gene therapy is currently under FDA review
How evolving medical therapies and more precise patient selection are reshaping the role of intestinal transplantation
Cleveland Clinic specialist discusses the new clinical practice guideline
Introducing Prajwal Rajappa, MD, MS, Director of Pediatric Precision Oncology
The one-stop THRIVE clinic addresses renal, psychosocial, educational and other challenges alongside cardiac surveillance
Study found lower rates of nausea, vomiting and healthcare use through 12 months of follow-up
What to know about the condition, and 5 strategies to improve patient care
Biologic therapy and advances in monitoring may ease treatment and follow-up for families