Leaders from Cleveland Clinic’s Hearing Implant team provide insight

By Sarah A. Sydlowski, AuD, PhD; Erika Woodson, MD; and Samantha Anne, MD
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
Candidacy criteria for cochlear implants (CI) are rapidly expanding. Patients who would have historically been dissuaded from considering CI are not only receiving them but experiencing some of the best outcomes. These former noncandidates demonstrate benefit not only in terms of speech recognition outcomes but also in terms of increased localization ability, improved quality of life and reduction of tinnitus.
Studies suggest that limiting CI to patients with bilateral severe-to-profound hearing loss is an outdated approach and may prevent some of the best candidates from benefiting from this life-changing technology. So compelling is the evidence that as of July 2019, Food and Drug Administration (FDA) labeling for one cochlear implant manufacturer now extends candidacy to patients with asymmetric hearing loss (AHL), up to and including single-sided deafness (SSD), which consists of one normal hearing ear.
Cleveland Clinic’s Hearing Implant Program has long been committed to improving access to cochlear implants for all potentially successful candidates, including those who were previously considered marginal or noncandidates by strict audiological criteria. As such, we have a rich history of compelling outcomes in adult patients with both AHL and SSD as well as more recently evolving experience with our pediatric population. We have based our decision to adopt a progressive approach to CI candidacy on the well-documented evidence that despite having a better, and in some cases, normal ear, AHL and SSD are potentially disabling conditions that cannot be equivalently remediated with other devices.
Advertisement
Even with normal contralateral hearing, individuals with SSD have difficulty localizing sound, hearing sounds arising from the deafened side and understanding speech in background noise. The challenges of SSD are perhaps intensified by the fact that unilateral hearing loss is a nearly invisible disorder1, with many communication partners unaware of the disability.
Individuals with SSD commonly report perceived hearing handicaps that often approach severe levels2 due to difficulty participating in group environments, inability to converse in lateralized communication settings such as when riding in the car, and general fatigue from increased listening effort, particularly when in challenging hearing environments. Many patients report social consequences that include feelings of embarrassment and isolation resulting from the inability to accurately follow conversations in a noisy environment.3
In children, any degree of unilateral hearing loss has been shown to lead to impaired educational progress including “higher rates of grade failure, higher incidence of individualized education programming (IEP), and increased need for educational resources and attention in the classroom”4 and to negative consequences for normal speech and language acquisition and development.5
In recent years, a series of clinical trials has demonstrated that CI in AHL or SSD may “help to restore binaural functionality, improve quality of life, facilitate reversal of neuroplasticity related to auditory deprivation,”4 effectively attenuate tinnitus, and improve localization and speech recognition ability.6,7,8 Most important, studies suggest that the addition of an electrically driven stimulus to normal contralateral hearing does not interfere with speech understanding capabilities and does not result in nonuse of the device. On the contrary, a systematic review of the literature reported ongoing device utilization by 96.5% of recipients.9
Advertisement
It is well documented that earlier implantation and shorter duration of deafness before implantation results in improved outcomes.10 Thus, identifying candidates who have suffered sudden sensorineural hearing loss without meaningful recovery can be among the most ideal candidates for CI. However, although we frequently observe positive outcomes in these populations, lack of FDA labeling has made it more challenging to obtain insurance approval for these patients.
As we continue to manage these newly labeled candidates, our program focuses on several key elements to optimize outcomes, as outlined in Table 1 below.
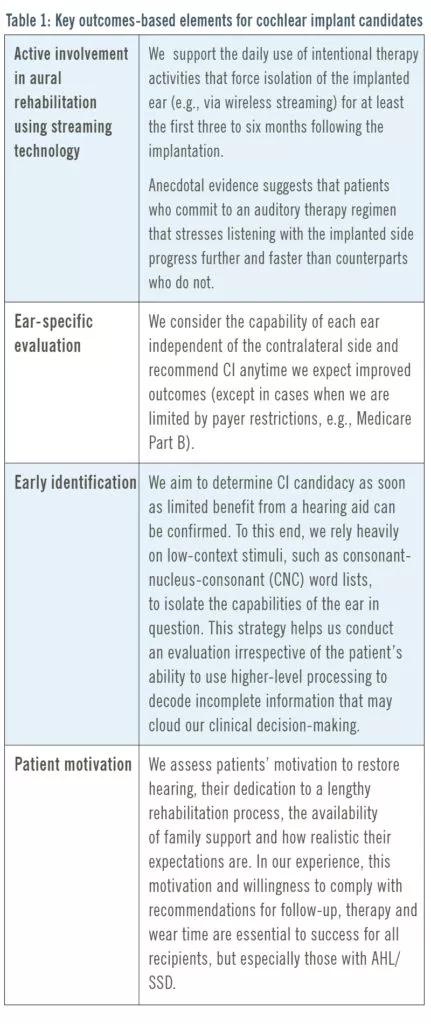
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/7c85f936-0469-4e19-b870-50a27c6d1e4e/19-ENT-4248-CochlearImplant-table1-800x1900-431x1024_jpg)
A 34-year-old female presented to Cleveland Clinic with a left temporal bone fracture from a fall eight years prior. Audiometric testing showed normal hearing in the right ear and a total profound sensorineural loss in the left. The patient had used a bone-anchored implant for approximately seven years but reported extremely limited benefit. In particular, she described loud, bothersome tinnitus (10/10 on tinnitus distress scale) and no localization capability. She had begun withdrawing from social situations and felt increasingly frustrated at her job (a busy office environment).
The patient completed several subjective questionnaires based on her experience (Figure 1). Her aided speech recognition capability was very poor (Figure 2). After she was approved for and received the left cochlear implant, she reported significantly improved subjective and objective assessments. Results reported at one-month post-activation are sustained at one-year post-activation. Notably, the patient reported a completely different experience than with her bone-anchored implant, remarking that the CI “has become a part of me.”
Advertisement
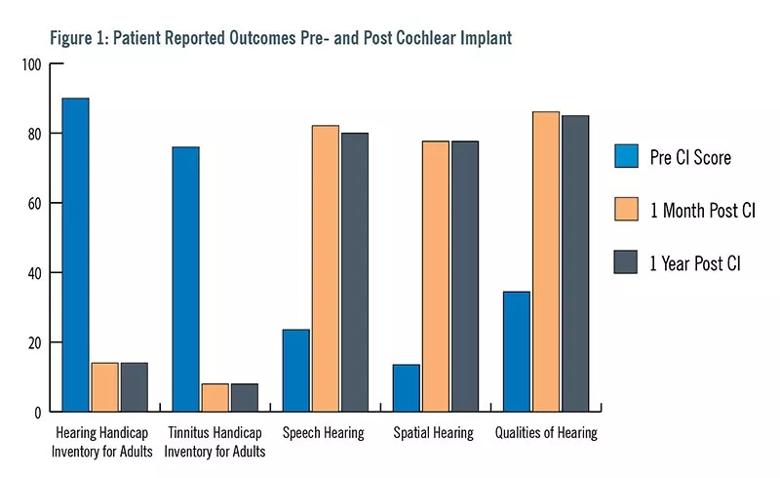
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/4d7c047a-8d7c-411b-8550-92365989a20d/19-ENT-4248-CochlearImplant-figure1-800x490_jpg)
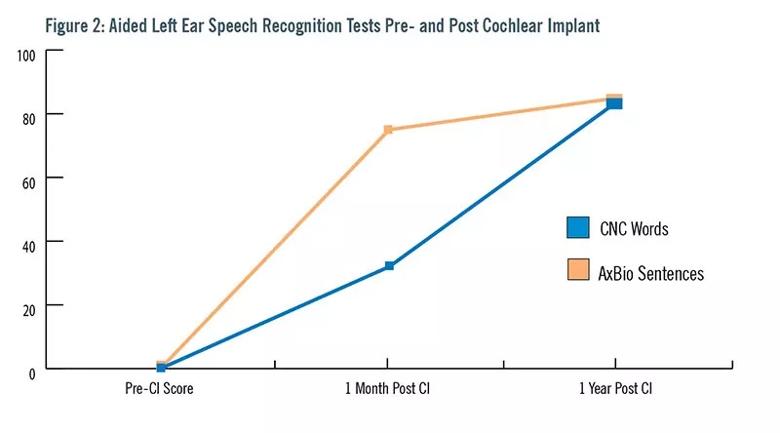
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/655ef834-6bfc-4ffe-a3ce-4d0db7e0f26c/19-ENT-4248-CochlearImplant-figure2-800x444_jpg)
A 4-year-old patient presented to Cleveland Clinic with bilateral hearing loss. The patient was born prematurely at 35 weeks due to an intrauterine growth restriction and spent two weeks in the NICU for breathing difficulty. Though she passed universal neonatal hearing screening at birth, by age 2 the family had concerns about her speech and language delay — specifically, poor expressive speech intelligibility.
At 3 years and 7 months, she underwent behavioral and electrophysiologic testing after the school psychologist raised concerns that her behavior was emblematic of hearing loss. Testing confirmed a moderately severe-to-profound sensorineural hearing loss in the right ear and a mild-to-severe sensorineural hearing loss in the left ear. She was fit with bilateral hearing aids, and aided speech recognition scores were obtained (Table 2).
Based on these results along with responses to parental questionnaires and communication evaluation, she was approved for a right cochlear implant, which was placed at age 4 years and 6 months. She continued use of a left hearing aid. By three months post-activation, her speech understanding had improved significantly on the same measures. By one year post-activation, her speech recognition scores (on more difficult, age-appropriate lists) were obtained (Table 3).
She is currently in a mainstream kindergarten classroom and participates in nearly monthly auditory-verbal therapy sessions during which highly intelligible, conversational speech has been noted.
Advertisement
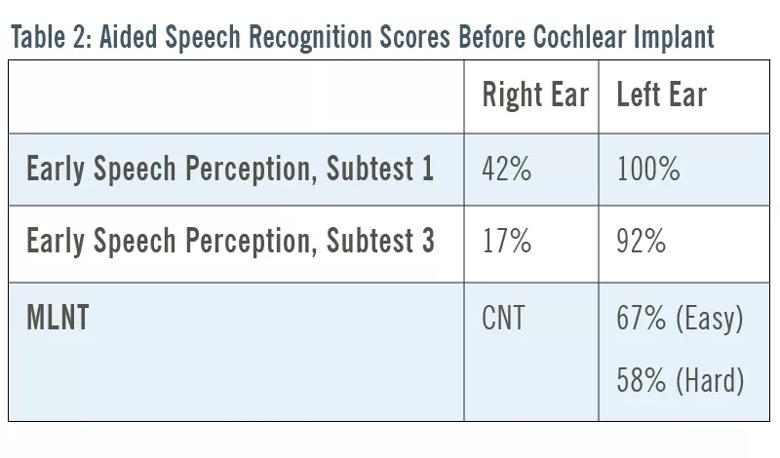
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/cc0d9dd3-9cab-426c-8969-dad0bacfa0fb/19-ENT-4248-CochlearImplant-table2-800x470_jpg)
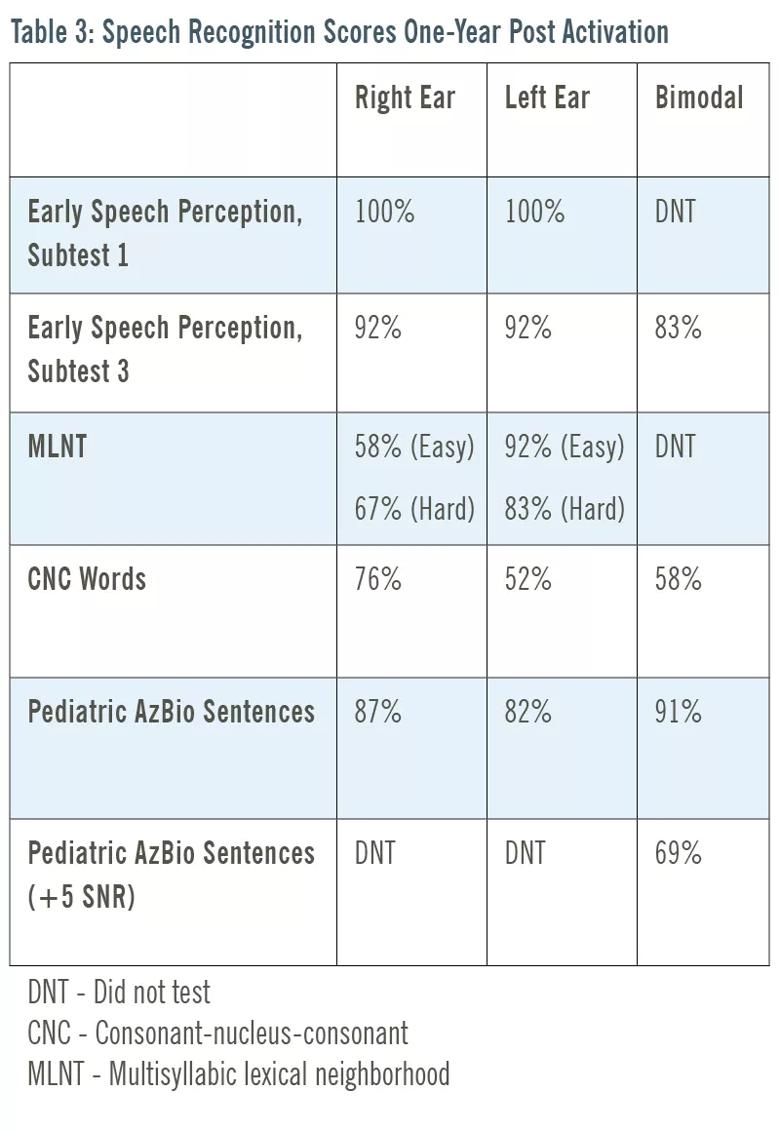
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/e9571d22-1b9c-4f63-855c-9fe7c5869bb1/19-ENT-4248-CochlearImplant-table3-800x1160_jpg)
1. Snapp H, Angeli S, Telischi FF, Fabry D. Postoperative validation of bone-anchored implants in the single-sided deafness population. Otology & Neurotology. 2012; 33(3): 291-296.
2 Newman CW, Jacobson GP, Hug GA & Sandridge SA. Perceived hearing handicap of patients with unilateral or mild hearing loss. Annals of Otology, Rhinology, & Laryngology. 1997; 106(3): 210-214.
3. Wie OB, Pripp AH, Tvete O. Unilateral deafness in adults: effects on communication and social interaction. Annals of Otology, Rhinology, & Laryngology. 2010; 119(11): 772-781.
4. Zeitler DM, Dorman, MF. Cochlear implantation for single-sided deafness: A new treatment paradigm. Journal of Neurological Surgery. 2019; 80, 178-186.
5. Anne S, Lieu JEC, Cohen MS. Speech and Language Consequences of Unilateral Hearing Loss: A Systematic Review. Otolaryngology-Head and Neck Surgery. 2017;157(4):572-579.
6. Arndt S, Aschendorff A, Laszig, R., et al. Comparison of pseudobinaural hearing to real binaural hearing rehabilitation after cochlear implantation in patients with unilateral deafness and tinnitus. Ear & Hearing. 2011; 32(1): 39-47.
7. Punte AK, Vermeire K, Hofkens A, De Bodt M, DeRidder D, et al. Cochlear implantation as a durable tinnitus treatment in single-sided deafness. Cochlear Implants International. 2011; 12, S26-S29.
8. Van de Heyning P, Vermeire K, Diebl M, Nopp P, Anderson I, et al. Incapacitating unilateral tinnitus in single-sided deafness treated by cochlear implantation. Annals of Otology, Rhinology, and Laryngology. 2008; 117, 645-652.
9. Vlastarakos PV, Nazos K, Tavoulari EF, Nikolopoulos TP. Cochlear implantation for single-sided deafness: the outcomes. An evidence-based approach. European Archives of Oto-Rhino-Laryngology. 2014; 271(8), 2119-2126.
10. Polonenko MJ, Papsin BC & Gordon KA. Delayed access to bilateral input alters cortical organization in children with asymmetric hearing. NeuroImage: Clinical. 2018; 17, 415-425.
Dr. Sydlowski is Audiology Director, Hearing Implant Program; Dr. Woodson is Section Head, Otology-Neurotology and Medical Director, Hearing Implant Program; Dr. Anne is Medical Director, Pediatric Ear and Hearing Disorders.
Advertisement

Analysis of HNSCC patients shows HPV status to be predictive of higher abundance of oncobacteria within the tumor

Case study illustrates the potential of a dual-subspecialist approach

Evidence-based recommendations for balancing cancer control with quality of life

Study shows no negative impact for individuals with better contralateral ear performance

HNS device offers new solution for those struggling with CPAP

Patient with cerebral palsy undergoes life-saving tumor resection

Specialists are increasingly relying on otolaryngologists for evaluation and treatment of the complex condition

Detailed surgical process uncovers extensive middle ear damage causing severe pain and pressure.