New study uses hyperglycemic clamp technique in individuals with idiopathic PAH
By Jacob Mey, PhD, RD, and Gustavo Heresi, MD, MS
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
Our group recently published a study that challenges the prevailing literature on insulin resistance in individuals with a rare disease, pulmonary arterial hypertension (PAH). These findings highlight the importance of metabolic research in PAH. We are hopeful this work will positively impact the field by promoting new lines of scientific inquiry, including both pharmacological and dietary approaches, for the treatment of PAH.
PAH is a rare, yet deadly chronic disease, characterized by remodeling of the pulmonary vascular bed leading to elevated pulmonary arterial pressure requiring progressive compensation by the right ventricle, and culminating in right heart failure. Despite recent clinical advances, prognosis remains poor with estimated three-year survival rates of 55% to 69% in newly diagnosed patients. The pathobiology of PAH is incompletely understood and none of the currently available treatments directly target the underlying pulmonary vascular remodeling or right heart dysfunction. As such, there remains a dire need to identify the underlying pathophysiology of PAH to improve disease management.
Recent literature suggests greater insulin resistance in PAH compared to healthy counterparts. However, these studies have primarily relied on static indices of insulin resistance (e.g. HOMA-IR) rather than dynamic measures from oral glucose tolerance tests (OGTT) or intravenous glucose clamp procedures. Our group first conducted OGTTs in PAH compared to age-, BMI- and sex-matched controls, verifying poor oral glucose tolerance in PAH, which coincided with reduced circulating insulin levels, suggesting a potential underlying issue of impaired pancreatic insulin secretion.1
Advertisement
Therefore, in our more recent study, we set out to investigate pancreatic insulin secretion in PAH using the gold-standard hyperglycemic clamp technique in individuals with idiopathic PAH and age-, BMI- and sex-matched controls. Similar to our OGTT results, individuals with PAH had reduced circulating insulin levels in response to IV-induced hyperglycemia, but surprisingly, pancreatic insulin secretion was similar to controls. We interrogated other metabolic tissues involved in glucose and insulin regulation suggesting
We also performed lipidomic analysis using an HPLC approach yielding an interesting finding; a bioactive lipid (palmitoleate) was significantly elevated in PAH and positively correlated with the degree of skeletal muscle insulin sensitivity. Palmitoleate has emerged in the literature as a potential insulin-sensitizing agent and our results support the continued investigation of palmitoleate in metabolic research.
Despite these findings, our results were limited by the small cohort (N =12) undergoing the hyperglycemic clamp procedures. Therefore, we conducted a larger-scale metabolomic analysis in 52 fasted subjects, mirroring our findings for fasted metabolism and the predominant upregulation of lipid and ketone metabolism in PAH. These metabolomics results provide additional confidence in the applicability of this research to the greater population of individuals with PAH.
Advertisement
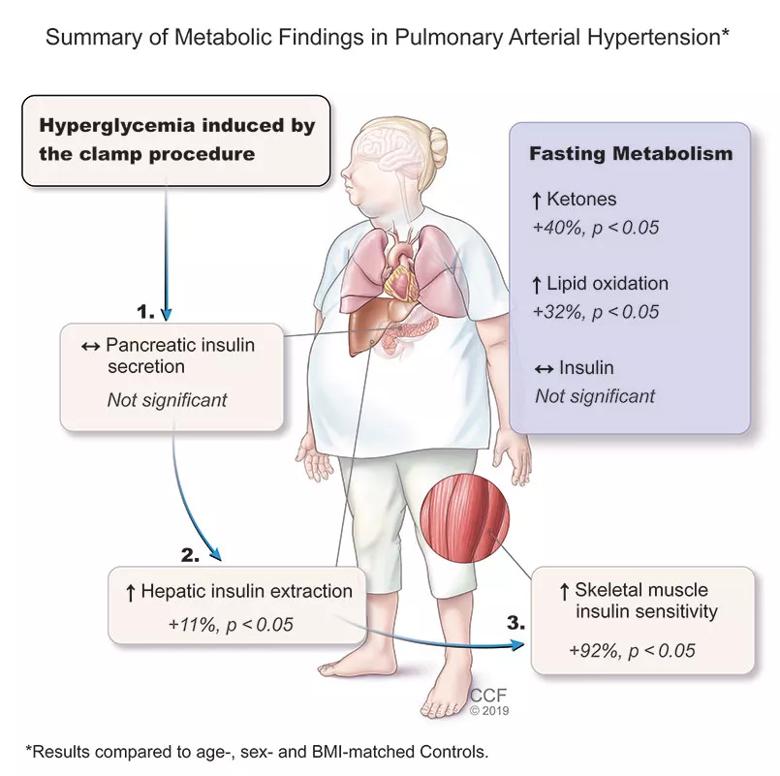
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/db3d0b4a-1bee-479c-a558-ba2f926d279d/805x-Inset-2020-Clamp-Abstract-Figure_jpg)
Reproduced with permission of the © ERS 2020: European Respiratory Journal 1901700; DOI: 10.1183/13993003.01700-2019 Published Early View 27 February 2020 – In Press
Taken together in the context of the literature, our data suggest that, rather than overt tissue insulin resistance in PAH, peripheral tissues in PAH are insulin sensitive, but some factors (possibly hepatic insulin extraction) are reducing the systemic insulin response to exogenous glucose (oral or intravenous). We hypothesize this may be due to the underlying PAH physiology which prefers to utilize ketone and lipid metabolism at the expense of glucose control (perhaps to compensate for the progressive heart failure, as ketones are a more efficient fuel source).
The next logical step is to test pharmacologic or lifestyle approaches to minimize glucose excursions and/or promote ketone and lipid metabolism for improving clinical outcomes in PAH. Our group is currently investigating a lifestyle intervention, involving aerobic exercise (60 min/day, 5 days/week, 12 weeks) and the Mediterranean diet in individuals with PAH.
Dr. Mey is a Postdoctoral Research Fellow at the Pennington Biomedical Research Center. His clinical interests include ketone bodies, skeletal muscle metabolism and malnutrition. Tweet him @CakeNutrition.
Dr. Heresi is Director of the Pulmonary Vascular and Chronic Thromboembolic Pulmonary Hypertension Program in the Department of Pulmonary and Critical Care Medicine, Respiratory Institute. His clinical interests include pulmonary hypertension, acute pulmonary embolism and chronic thromboembolic pulmonary hypertension (CTEPH). Tweet at him @heresi_gustavo
Advertisement
For inquiries related to this research, you can reach Dr. Heresi at 216.636.5327 or heresig@ccf.org.
Advertisement
Advertisement

Takeaways from the most recent annual meeting centered around clinical advances, AI integration and professional development

Recent breakthroughs have brought attention to a previously overlooked condition

A review of treatment options for patients who may not qualify for surgery

Looking at the real-world impact and the future pipeline of targeted therapies

The progressive training program aims to help clinicians improve patient care

New breakthroughs are shaping the future of COPD management and offering hope for challenging cases

Exploring the impact of chronic cough from daily life to innovative medical solutions

How Cleveland Clinic transformed a single ultrasound machine into a cutting-edge, hospital-wide POCUS program