New study finds that the average patient can expect to lose an additional 3.5kg on canagliflozin
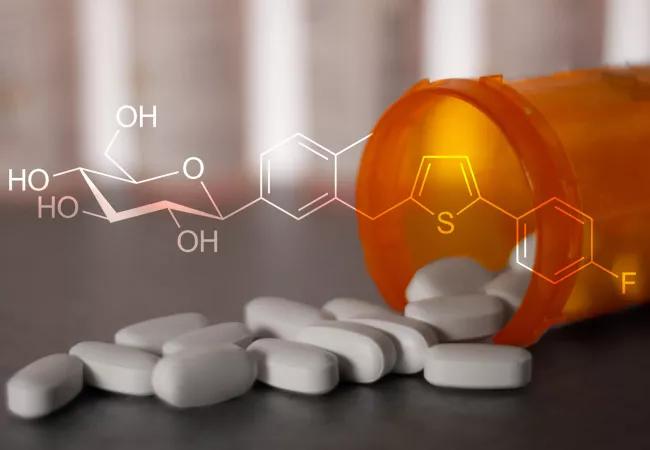
Canagliflozin is useful in reducing body mass index and blood glucose levels after metabolic surgery in patients with type 2 diabetes (T2D), a recent clinical trial finds. These changes may result in decreases in cardiovascular disease risk. The trial results were presented at the 2019 Annual Meeting of the American Association of Clinical Endocrinologists in Los Angeles.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
“Prior to this study, it had been suggested that canagliflozin might be prescribed concurrently with metabolic surgery, but there wasn’t a lot of evidence to support the recommendation,” says Sangeeta Kashyap, MD, a physician in the Cleveland Clinic Endocrinology & Metabolism Institute and senior author of the abstract.
Metabolic surgery is gaining popularity in the treatment of obesity and T2D, as it has profoundly positive effects on weight loss and blood glucose control. Although metabolic surgery often helps patients achieve remission from T2D, glycemic control begins to degrade two years after surgery for some patients. This relapse may parallel weight regain.
This double-blind, prospective study sought to determine the effects of canagliflozin, a renal sodium glucose-cotransporter-2 inhibitor (SGLT2) on weight loss and blood glucose levels two or more years after metabolic surgery. In order to be eligible for the study, participants were required to be at least one year—but not more than 15 years—out from their metabolic surgery (i.e., either adjustable gastric banding, Roux-en-Y gastric bypass, or sleeve gastrectomy) at screening. Participants also needed to be 20-75 years of age, with a diagnosis of T2D before and after weight loss surgery, have an HbA1C of >6.5% but <11%, and an eGFR of >60mL/min.
After screening and an eight-week possible wash-out period for any other antidiabetic medications, participants (N = 16) were randomized into either canagliflozin (N = 11) or placebo groups (N = 5). There were six visits associated with the trial (at screening, baseline, randomization, six weeks, three months and six months). Following randomization, subjects in the canagliflozin group received 100 mg/day for two weeks, and then were titrated to 300 mg/day as tolerated.
Advertisement
Participants in the canagliflozin group experienced significant reductions in BMI, body fat composition and fasting glucose levels. On average in this population, BMI decreased 2.89 kg/m2, android fat decreased 3% and truncal fat decreased 2.67%. On average, fasting blood glucose decreased 32.90 mg/dL in the canagliflozin group and increased 11.71 mg/dL in the placebo group.
“Our findings indicate that patients can expect an average weight loss of 3.5 kg on canagliflozin, while maintaining lean mass,” notes Dr. Kashyap. “In fact, the weight loss achieved in only six months of canagliflozin is comparable to two years on liraglutide. Additionally, a reduction in visceral adiposity is associated with insulin resistance and lowers the risk of cardiovascular disease.”
Initial studies found reductions in bone mineral density (BMD) in patients taking canagliflozin; however, more recent studies find no increase in fracture risk compared to glucagon-like peptide-1 (GLP-1) receptor agonists. In this trial, minimal changes in BMD were noted, though the small sample size and short duration of the study may have impacted those results.
“For patients who are seeing their weight loss plateau following metabolic surgery and who have inadequately controlled glycemic levels, canagliflozin may support further weight loss and a subsequent reduction in risk factors for cardiovascular disease,” Dr. Kashyap states.
Future research might focus on identifying a larger cohort of eligible subjects, as well as comparing the impact of other antidiabetic medications —in addition to the placebo—on weight loss and glycemic control.
Advertisement
Advertisement

A weight-management program plus anti-obesity medication performs well

TRANSITION-T2D RCT results for patients with T2D receiving MDI

Incidence, outcomes and management

Cleveland Clinic endocrinologists work to identify protocols for improving care

Cleveland Clinic’s Endocrinology & Metabolism Institute is working to offer novel opportunities and therapies for diabetes and kidney disease

New findings indicate the importance of achieving sufficient sleep levels in regard to personal glucose targets
A recent study evaluates the correlation between preoperative calcium and parathyroid hormone values as predictors of gland volume and multi-gland disease

A look at how to asses hCG elevation