Stress and immunosuppression can trigger reactivation of latent virus
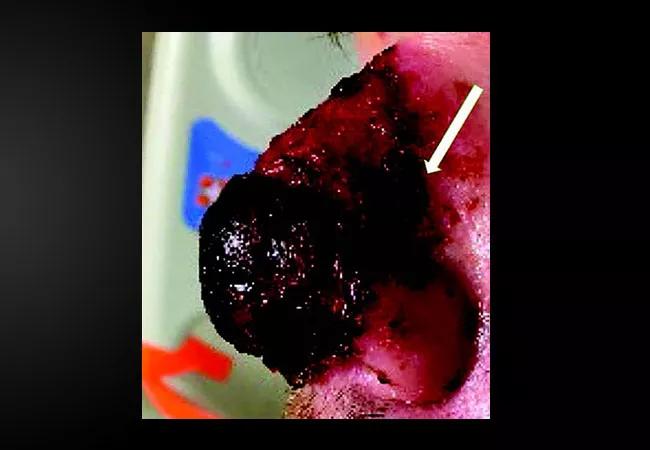
Above: Herpes simplex virus infection of the nose. A shave biopsy of the lateral border of the ulcerated plaque (arrow) revealed ulceration with spongiosis, serous crust and a lymphohistiocytic infiltrate.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
Editor’s Note: This article was originally published in the Cleveland Clinic Journal of Medicine.
By Nina L. Tamashunas, MD, Robert Simonds, MD, and Sarah Young, MD
A 37-year-old man with epilepsy presented to the emergency department with a one-week history of skin changes of the nose that began after being scratched on the nose. A few days after the scratch, he hit the tip of his nose on steel bundles during a seizure. He then developed a headache, and his nose began to swell and develop a black appearance at the tip. He had sought attention at another hospital, where he was prescribed doxycycline out of concern for a skin and soft-tissue infection.
At our emergency department, physical examination revealed hemorrhagic crusting, ulceration and black eschar with peripheral erythema of the tip of the nose and extending upward to the dorsal surface, prompting concern for extensive nasal skin necrosis. Intravenous vancomycin, clindamycin and piperacillin-tazobactam were started empirically.
Laboratory testing demonstrated only an elevated C-reactive protein, and blood cultures and lumbar puncture were negative. Head, facial and sinus computed tomography revealed soft-tissue edema without bony erosion. Nasal endoscopy showed healthy nasal mucosa.
Shave biopsy demonstrated ulceration with spongiosis, serous crust and a lymphohistiocytic infiltrate. Tissue culture was negative for fungi and acid-fast bacilli, but positive for group B Streptococcus. Polymerase chain-reaction testing was negative for herpes simplex virus type 1 (HSV-1) and varicella-zoster virus, but positive for HSV-2. Therefore, the patient was continued on vancomycin (total of seven days) and started on acyclovir (for two days) during hospitalization, and was discharged with a 10-day course of oral valacyclovir 1 g twice daily and amoxicillin-clavulanate. The patient’s nose continued to improve on this therapeutic regimen as an outpatient.
Advertisement
The differential diagnosis for this patient’s presentation included infections such as HSV-1, HSV-2, Chikungunya virus, invasive fungal infections, rhinoscleroma, Hansen disease (formerly leprosy), and Mycobacterium tuberculosis; inflammatory conditions such as granulomatosis with polyangiitis; and neoplasms such as natural killer or T-cell lymphomas.
In this patient, hemorrhagic ulceration, immunocompetence, limited comorbidities, and no known exposure to uncommon infectious agents served as clues for a diagnosis of HSV infection. HSV infection commonly presents as vesicles or ulcers, which may represent deroofed vesicles. Additionally, ulcers may have an overlying yellow crust due to serous exudate from the disrupted vesicles or from secondary impetiginization.
Skin lesions caused by invasive fungal infection can be the result of a localized or disseminated process and are often characterized by central necrosis. These infections commonly affect patients with underlying immunosuppression (e.g., human immunodeficiency virus infection, hematologic malignancy, glucocorticoid use) or diabetes mellitus. Lupus vulgaris, a cutaneous form of mycobacterial infection, usually presents on the head or neck in adults in a variety of ways including plaque, ulcerative, vegetative, papulonodular and tumorlike forms, but lesions tend to be chronic and progressive in nature.
HSV infection may present as gingivostomatitis, eczema herpeticum, erythema multiforme, herpetic whitlow, genital ulcerations, keratitis, chorioretinitis, encephalitis, Bell palsy, esophagitis and hepatitis. It can be responsible for primary, latent and recurrent infections, as it is dormant in the dorsal root ganglion of sensory neurons. Potential triggers for reactivation include physical stress, psychological stress and immunosuppression.
Advertisement
HSV is most commonly found in the oral mucosa but has also been detected in tears and nasal mucosa of immunocompetent adults.1 Cutaneous or mucosal HSV can be assessed with polymerase chain reaction testing or culture of swabs of the base of the ulcerative lesions. As in this patient, vesiculation and ulceration of the dorsal surface of the nose without intranasal involvement due to HSV infection has been reported in an immunocompetent individual with a remote history of trauma to the nose.2 Intranasal HSV infection with and without evidence of necrosis has been reported in immunocompromised individuals, and this presentation is frequently mistaken for invasive fungal rhinosinusitis.3,4 Management with systemic antiviral therapy is effective, and surgical debridement is required only in severe cases.
Dr. Tamashunas is an Internal Medicine Resident at MetroHealth in Cleveland, Ohio. Dr. Simonds is a Fellow in Cleveland Clinic’s Department of Dermatology. Dr. Young is Associate Staff in Cleveland Clinic’s Dermatology & Plastic Surgery Institute.
References
1. Ramchandani M, Kong M, Tronstein E, et al. Herpes simplex virus type 1 shedding in tears and nasal and oral mucosa of healthy adults. Sex Transm Dis 2016; 43(12):756–760. doi:10.1097/OLQ.0000000000000522.
2. Powell HR, Almeyda J. An immunocompetent patient presenting with severe nasal herpes simplex: a case report. Cases J 2009; 2:9079. doi:10.1186/1757-1626-2-9079.
3. Kaplon A, Kedarisetty S, McLean C. Intranasal herpes simplex virus (HSV): a unique mimicker of acute invasive fungal rhinosinusitis. Ear Nose Throat J 2021; 100(5_suppl):498S–499S. doi:10.1177/0145561319885715
Advertisement
4. Grimmer JF, Zhou H, Vanderhooft SL. Necrotizing herpes simplex infection of the nose. Otolaryngol Head Neck Surg 2007; 137(4): 689–690. doi:10.1016/j.otohns.2006.11.035.
Advertisement
Advertisement
Family history may eclipse sun exposure in some cases
Consider secondary syphilis in the differential of annular lesions
Persistent rectal pain leads to diffuse pustules

Two cases — both tremendously different in their level of complexity — illustrate the core principles of nasal reconstruction

Low-dose, monitored prescription therapy demonstrates success

Antioxidants, barrier-enhancing agents can improve thinning hair
A case report of a thumbnail deformity