How the technique could yield an atlas of functional brain connections
By Dileep Nair, MD, and John Mosher, PhD
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
In 2004, Cleveland Clinic’s Epilepsy Center published the first of several articles based on a technique — cortico-cortical evoked potentials (CCEPs) — pioneered by our neurophysiology lab using low-frequency electrical stimulation of electrodes implanted in patients undergoing invasive monitoring for epilepsy surgery.1,2 Low-frequency cortical stimulation (1 Hz) is used in CCEP recordings to determine which other brain regions respond by observing a measurable evoked signal in distant or nearby cortical regions (Figure 1). This technique has now been widely used in several other epilepsy centers across the world after our initial publications.
The CCEP methodology allows in vivo access to direct and indirect interactions across various nodes in the human brain. Our most recent report, published in Brain in 2017,3 shows correlation of these measures with the epileptic networks assessed using ictal single-photon emission computed tomography (SPECT).
Based on the strength of CCEP research findings, our center has received an R01 grant from the National Institute of Neurological Disorders and Stroke. The five-year grant is supporting a study to develop a brain map of CCEP responses from across hundreds of patients who have undergone epilepsy surgery using the invasive technique of stereoelectroencephalography (SEEG). The goal is to create a brain map of connectivity where all the implanted electrodes are nonlinearly co-registered into a single brain template. In this way, greater sampling of regions of brain will become available so that strengths of connections from various nodes of stimulation can be created such as those shown in the circle map in the series of images below.
Advertisement
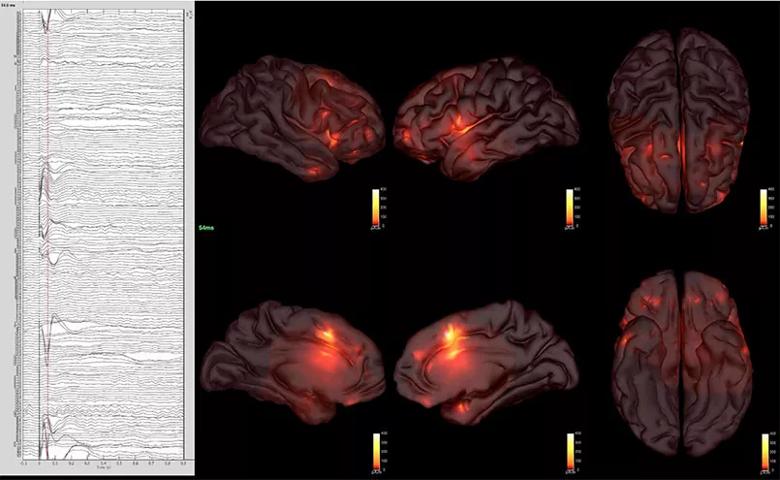
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/13df03d7-3a87-4260-9033-85e7ddac1669/17-NEU-4469-Nair-Mosher-CCEP-inset1_jpg)
A patient’s CCEP responses are depicted as waveforms on the left with response estimates depicted by scaled colors in the MRI scans on the right. Brain currents were estimated using a “minimum energy” constraint that keeps the currents to a minimum in the vicinity of the electrodes to generate a working estimate for use in studying brain dynamics.
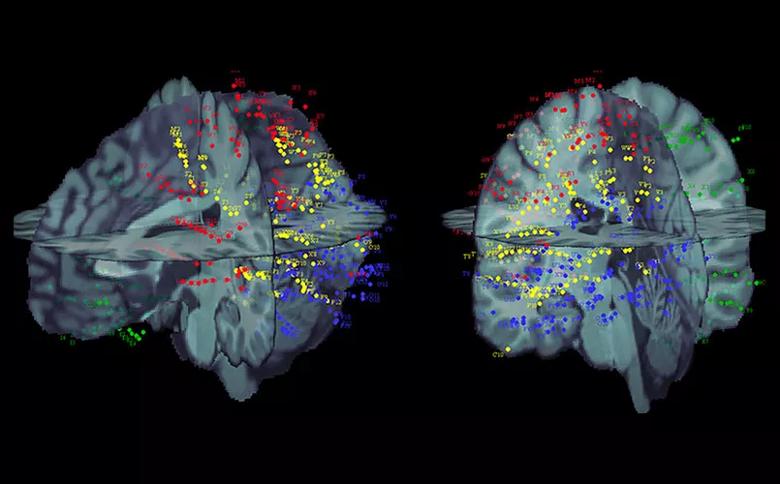
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/fdf4d805-61cd-40a7-bfe2-26f78d4ef6d8/17-NEU-4469-Nair-Mosher-CCEP-inset2_jpg)
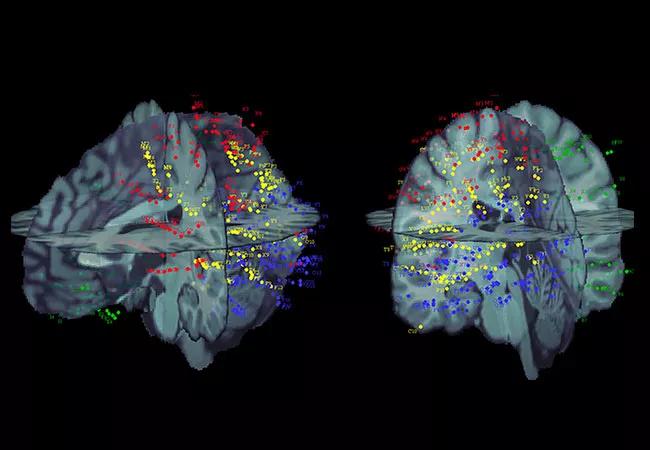
SEEG contacts from four patients (each shown with a different color) warped to a common atlas using open-source Brainstorm software.
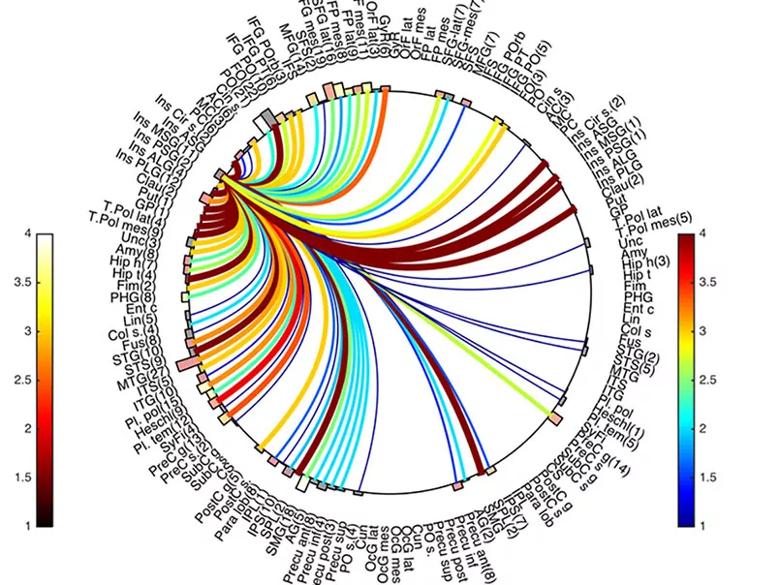
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/e4353ffc-a024-44a5-84fc-bbdd9651c7f7/17-NEU-4469-Nair-Mosher-CCEP-inset3_jpg)
Circle map showing example connectivity from one region to other regions of interest for six patients, calculated using RMS of the response at each contact.
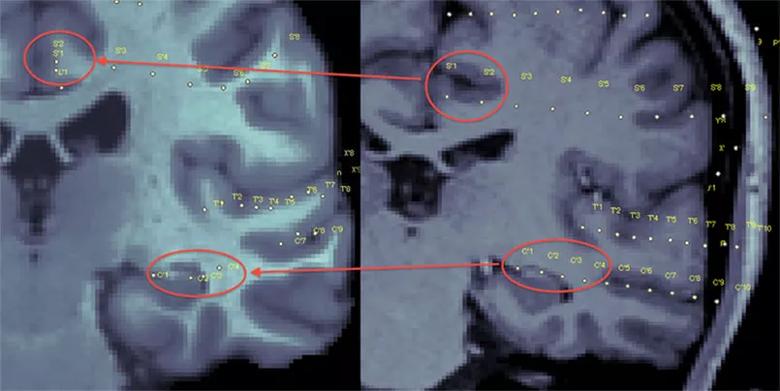
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/23741b2b-9012-419a-961c-4f484e980f05/17-NEU-4469-Nair-Mosher-CCEP-inset4_jpg)
Nonlinear registration of contacts from patient to the common atlas.
Our hope is that this information will give us better insight into how to craft more accurate surgical resections in patients with nonlesional medically intractable focal epilepsy if we can uncover the primary epileptic node noninvasively. The overarching goal of this research is to develop a brain atlas of functional connections between various areas of the human brain. This atlas would serve as a guide for mapping physiological and pathological networks involved in focal epilepsies and various neurological and psychiatric diseases.
Advertisement
Dr. Nair is Section Head of Adult Epilepsy in Cleveland Clinic’s Epilepsy Center. Dr. Mosher is a research scientist in the Epilepsy Center.
Advertisement
Advertisement

Recommendations on identifying and managing neurodevelopmental and related challenges

Phase 2 trials investigate sitagliptin and methimazole as adjuvant therapies

Aim is for use with clinician oversight to make screening safer and more efficient

Rapid innovation is shaping the deep brain stimulation landscape

Study shows short-term behavioral training can yield objective and subjective gains

How we’re efficiently educating patients and care partners about treatment goals, logistics, risks and benefits

An expert’s take on evolving challenges, treatments and responsibilities through early adulthood

Comorbidities and medical complexity underlie far more deaths than SUDEP does