Review paper offers three-step treatment guidelines

For most of the half million Americans who live with chronic urticaria, the cause of the condition isn’t known. Yet, today there are quite a few treatment options that help minimize the condition’s distressing symptoms.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
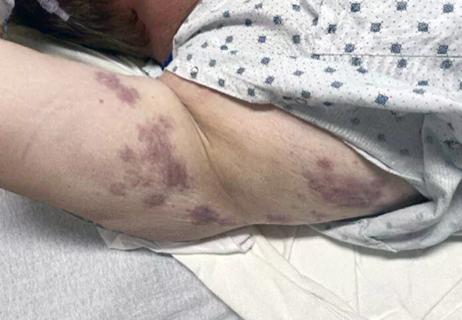
About 0.23% of the population has chronic urticaria, defined by the continuous or intermittent presence of wheals and/or angioedema for at least six weeks. The condition most frequently affects females over the age of 40 years, although it can appear at any age, and in both sexes. Unlike acute urticaria, which more frequently is triggered by a drug, food, or infection, chronic urticaria is commonly idiopathic.
Chronic urticaria can profoundly impair quality of life, with random episodes of intense itching that disrupt sleep and interfere with physical, social, and emotional functioning.
“Chronic urticaria is frequently associated with sleep disruption and significant impairment in quality of life. Even though we typically can’t answer the question commonly posed by patients, as we are unable to identify a cause, we can do a lot to help them with treatment that can lead to tremendous improvements in quality of life,” says David Lang, MD, who recently published a review paper on chronic urticaria in the New England Journal of Medicine.
Dr. Lang is Professor of Medicine at Cleveland Clinic’s Lerner College of Medicine and the former chair of the Department of Allergy and Clinical Immunology, one of the largest allergy/immunology programs in the United States with more than 20 staff members. In the paper, he provides a summary of stepped-care treatment synthesized from current evidence-based allergy-immunology and dermatology guidelines:
Step 1: A comprehensive history, assessment of baseline patient-reported outcomes, and a complete physical examination should be done initially, but laboratory testing should be limited. “There is no need to do extensive laboratory testing – this is not cost-effective and evidence indicates that will not lead to improved outcomes,” Dr. Lang says.
Advertisement
Validated tools for assessing patient-reported outcomes include the urticaria and angioedema activity scores, the urticaria and angioedema control tests, and the urticaria and angioedema quality-of-life scores. “It’s very important for providers to use patient-reported outcomes for initial and follow-up visits,” Dr. Lang emphasizes.
Patients should be counseled on appropriate avoidance measures and lifestyle changes, particularly if chronic inducible urticaria syndrome is present.
First-line pharmacotherapy is a second-generation H1-antihistamine as monotherapy, such as cetirizine (brand name Zyrtec), desloratadine (Clarinex), fexofenadine (Allegra), levocetirizine (Xyzal) or loratadine (Claritin). “These are equally effective as the 1st generation antihistamines, like diphenhydramine [Benadryl] or hydroxyzine [Atarax], without the untoward effects of sedation and anticholinergic effects,” Dr. Lang advises.
Step 2: If the patient is still experiencing symptoms – which will be the case for more than half – the dose of the second-generation H1-antihistamine can be escalated beyond that approved by the US Food and Drug Administration (FDA), a step that has been shown to improve symptom control without dramatically increasing side effects.
“The best next step in management if a patient doesn’t respond to the 2nd generation antihistamine at FDA-approved dosing is to increase the dose, as tolerated, up to fourfold,” Dr. Lang says.
Adjunctive therapies such as H2 antihistamines or antileukotrienes may also be helpful in combination with second-generation H1 antihistamines.
Advertisement
Step 3: For patients in whom control isn’t achieved with Step 2, a trial of omalizumab (Xolair) would come next. “For the treatment of refractory patients, there are high-quality data demonstrating the efficacy of omalizumab,” Dr. Lang says, noting the biologic agent was first FDA-approved for treating refractory allergic asthma in 2003 and subsequently for antihistamine-resistant chronic urticaria in 2014. Since then, “it has improved the landscape so that a substantial proportion of antihistamine-resistant patients become well controlled with the administration of omalizumab.”
But if omalizumab doesn’t work, alternatives include cyclosporin (Sandimmune), hydroxychloroquine, sulfasalazine, mycophenolate (CellCept), stanozolol and colchicine.
Oral glucocorticoids can be used to treat flares and restore control, but these medications should be regarded as short-term agents and are generally not prescribed on a regular basis due to the potential for untoward effects associated with long-term use.
Many more agents are in development for the treatment of chronic urticaria. Clinical trials are underway for anti–inter leukin-4/interleukin-13 (ClinicalTrials.gov number, NCT04180488), anti–interleukin-5 (NCT03494881 and NCT04612725), anti–thymic stromal lymphopoietin (NCT04833855), and anti–Siglec-8 agents (NCT03436797); more potent anti-IgE monoclonal antibodies (NCT03580356); and Bruton’s tyrosine kinase inhibitor therapies (NCT05030311).
What’s needed now, Dr. Lang says, “are validated biomarkers that can tell us which medication will or won’t work. We also need biomarkers that have prognostic significance for the severity and duration of the course of chronic urticaria. Currently, there’s no way to know. Hopefully, that will change.”
Advertisement
Advertisement
Symptoms complement one another

Recent breakthroughs have brought attention to a previously overlooked condition

A review of treatment options for patients who may not qualify for surgery

Looking at the real-world impact and the future pipeline of targeted therapies

The progressive training program aims to help clinicians improve patient care

New breakthroughs are shaping the future of COPD management and offering hope for challenging cases

Exploring the impact of chronic cough from daily life to innovative medical solutions

How Cleveland Clinic transformed a single ultrasound machine into a cutting-edge, hospital-wide POCUS program