A complex case establishes the utility of long-term ECMO in oncology
Extracorporeal membrane oxygenation (ECMO), delivered over the course of 40 days, proved to be a lifesaving intervention in an 18-year old man with primary mediastinal B-cell lymphoma invading into the trachea. While on ECMO, the patient successfully underwent fluorodeoxyglucose positron emission tomography (FDG-PET) imaging and treatment with radiation and chemotherapy, and eventually fully recovered. The case study, published in the May 2020 issue of Pediatric Blood & Cancer, established for the first time the utility of long-term ECMO in oncology.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
“There really are no reports in the literature of cancer patients receiving chemotherapy on ECMO for this long of a duration,” says Cleveland Clinic Children’s pediatric oncologist Seth J. Rotz, MD, study corresponding author. “Giving chemotherapy on ECMO is very difficult to do, let alone do it for 40 days at a time.” He further notes that this was the first time a PET scan was performed on a patient on ECMO.
The patient presented to the Cleveland Clinic emergency department with worsening respiratory distress. A chest X-ray revealed a large mediastinal mass, which was subsequently confirmed as primary mediastinal B-Cell Lymphoma on biopsy. After undergoing a series of procedures including airway stenting, intubation and several bronchoscopies, the patient was treated with cyclophosphamide, doxorubicin, vincristine, prednisone and rituximab. However, the tracheal stent migrated and became stuck to the granulation tissue, causing major hemorrhage and requiring him to undergo cardiopulmonary resuscitation with simultaneous initiation of ECMO. Figure 1 highlights the timeline of major clinical events and Figure 2 shows the images obtained during treatment.
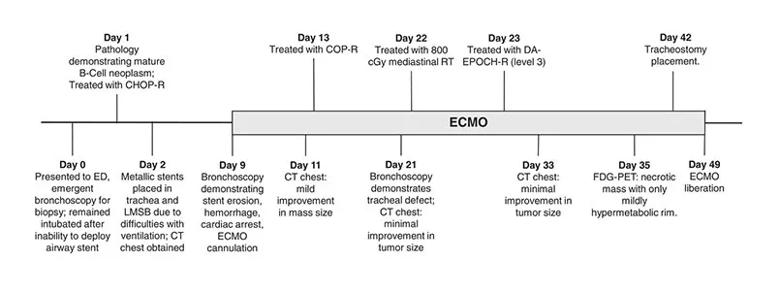
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/a4bf07f7-1d96-4dd7-b8d3-8b2ad1a576d3/805x-Inset-1-ECMO_jpg)
Timeline of clinical events. Note: LMSB, left main stem bronchus; COP-R, cyclophosphamide, vincristine, prednisone, and rituximab.*
“The patient was in the bronchoscopy suite having an airway stent adjusted when he started to bleed. His airway quickly filled with blood, so he was essentially drowning in his own blood,” says Cleveland Clinic thoracic surgeon Alejandro Bribriesco, MD, ECMO expert and part of the patient’s multidisciplinary care team. “I received the emergent call to activate our Respiratory ECMO team. We ran down to the bronchoscopy suite and as we were setting up, the patient’s oxygen levels dropped so low that his heart temporarily stopped and so we quickly initiated him on venoarterial ECMO.”
Advertisement
The patient was converted to venovenous ECMO, which took over the function of his lungs, throughout additional cycles of treatment with chemotherapy and radiation. In addition to receiving cancer treatment, he was also treated for a series of potentially life-threatening complications while on ECMO. Dr. Bribriesco explains that a fortunate circumstance in this case was that the patient’s lungs were not significantly damaged by the tumor.
“His lungs themselves were always okay,” he says. “His airway was just completely compressed and obstructed so air could not effectively get in or out of his lungs. But as the tumor shrunk, he was able to better move air through the otherwise normal lungs so that we were able to safely separate him from the ECMO machine.”
After a little over 5 months in the hospital the patient was released and continues to do well.
“He’s taking college classes and doing amazingly well,” says Dr. Rotz. “He finished therapy at the end of the prescribed treatment course, and we continue to get CAT scans of his chest every 3 months or so, but he is not getting any cancer-specific therapy. He is still being treated for a couple of treatment complications, but those are fairly minor in the context of this case.”
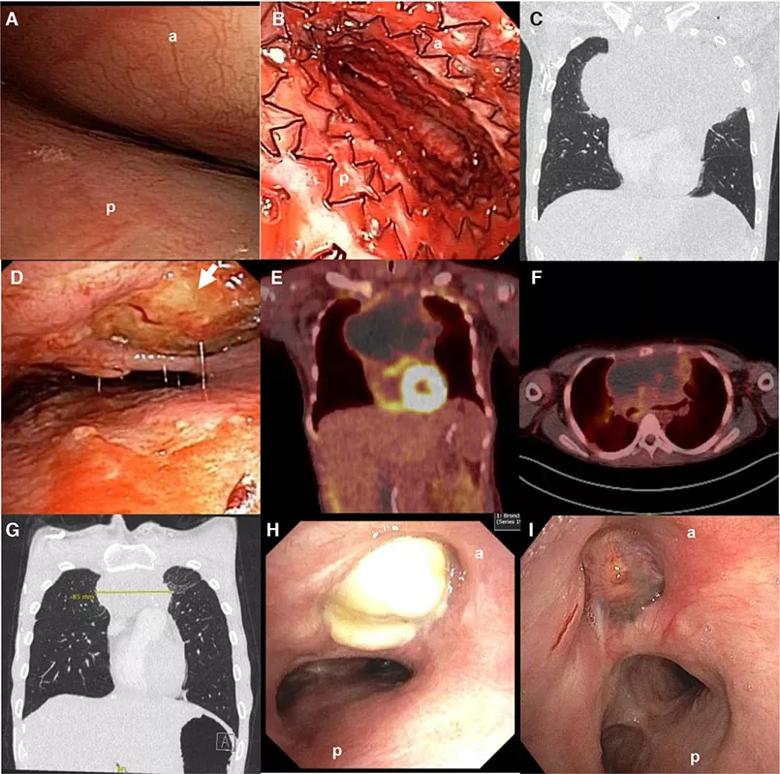
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/02a9607b-dfe9-4194-94ef-a85963f523a3/805x-Inset-2-ECMO_jpg)
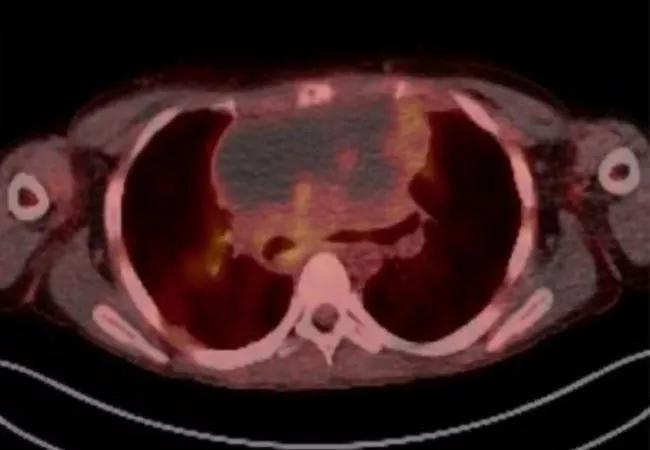
A, Initial bronchoscopic view of the very distal trachea with inability to see the main carina or main stem bronchi. B, Distal trachea metallic stent deployment showing mild improvement in airway patency but still with significant stenosis. C, Initial CT chest demonstrated an 18 × 12 × 16 cm anterior mediastinal mass. CT imaging also demonstrated a 3 cm right renal pole mass, and near complete effacement of the superior vena cava with extensive collateral formation (not shown).D, Bronchoscopic view of distal trachea on ED 12/HD 21 demonstrating mildly improved compression, but the anterior mediastinal mass invading through the trachea (arrow). The erosion remained essentially sealed throughout treatment as no pneumomediastinum developed. E, Positron emission tomography (PET) scan performed while on extracorporeal membrane oxygenation (ECMO) demonstrating a mostly necrotic tumor with mildly hypermetabolic ring surrounding the periphery. F, PET scan at the end of therapy demonstrating fluorodeoxyglucose (FDG) avidity in the periphery (Deauville 4). A transbronchial biopsy was obtained of an FDG avid portion of themass and demonstrated cellular debris and necrosis. Several subsequent PET scans continued to show mildly hypermetabolic areas, thought to be due to macrophages breaking down residual necrotic tumor; tumor dimensions continued to decrease on subsequent scans. G, Residualmass on CT scan at 9 months after the completion of final course of dose-adjusted etoposide, prednisone, vincristine, cyclophosphamide, doxorubicin, and rituximab (DA-EPOCH-R). H, Airway view of patient’s distal trachea and residual mass immediately after the completion of final cycle of chemotherapy. (I) and at 9 months post-therapy with re-epithelialization. Note: (a) anterior trachea; (b) posterior trachea.*
Advertisement
Teamwork and inter-departmental collaboration, as well as timely responses to the complications that arose during treatment, were essential in securing a positive outcome for the patient, Dr. Bribriesco says.
“We are all very fortunate to work at a place like the Cleveland Clinic where we have expertise in so many different areas as well as teams immediately available at all times, ready to respond,” he says. “I remember very vividly getting the page around 5 or 6 pm on a Friday night….thankfully, the OR and perfusion teams were available to come together and mobilize across the hospital so quickly on a Friday night. We got there just in time to get him on ECMO machine before he was beyond saving.”
Experts from more than 10 different departments were involved in the patient’s care during the course of his stay at Cleveland Clinic, including pulmonary medicine, critical care, interventional pulmonary care, infectious disease, palliative care, thoracic surgery, cardiac anesthesia, neurology, radiation oncology, respiratory therapy, physical therapy, pharmacy and nursing. Furthermore, the patient’s care team had to take special precautions when transporting him from the Cardiothoracic ICU to the Taussig Cancer Center where he received radiotherapy.
“There was a lot of planning that went into this — from the nursing side, the ICU side, the surgery side and the perfusion side — we had to precisely map the ¾ mile route across the skywalk between hospital buildings he would have to take,” says Dr. Bribriesco. “I was told that this was only the second time that we’ve closed down the skywalk between buildings. The only other time was when President Obama was here.”
Advertisement
“As we were coming up with this plan, people weren’t saying it can’t be done, but rather, they were all asking how they could help to make it happen,” he adds. ”So, it is this idea of putting patients first, really thinking outside of the box to find unique solutions to seemingly impossible problems that is a testament to the spirit that’s here at Cleveland Clinic.”
*Note: Images used with permission. Originally published in Rotz SJ, Almeida FA, Koyfman S, et al. Continuous infusion chemotherapy, radiotherapy, and FDG-PET are feasible during exracorporeal membrane oxygenation. Pediatr Blood Cancer. 2020 Jul;e28429. Epub ahead of print.
Advertisement
Advertisement

Combining advanced imaging with targeted therapy in prostate cancer and neuroendocrine tumors

Early results show strong clinical benefit rates

The shifting role of cell therapy and steroids in the relapsed/refractory setting

Radiation therapy helped shrink hand nodules and improve functionality

Standard of care is linked to better outcomes, but disease recurrence and other risk factors often drive alternative approaches

Phase 1 study demonstrates immune response in three quarters of patients with triple-negative breast cancer

Multidisciplinary teams bring pathological and clinical expertise

Genetic variants exist irrespective of family history or other contributing factors