Glycation lowers ApoA1 stability, destroys HDL in T2DM
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/08dcbdf5-4ccd-4405-b313-2fa198632b61/Cholesterol-2-jpg)
Cholesterol
A Cleveland Clinic study for the first time revealed a mechanism that rapidly destroys high-density lipoprotein (HDL) in people with Type 2 diabetes, negating cardiovascular protections of the so-called good cholesterol.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
Sangeeta Kashyap, MD, in Cleveland Clinic’s Endocrinology & Metabolism Institute, and co-author Jonathan D. Smith, PhD, the Geoffrey Gund Endowed Chair for Cardiovascular Research in the Department of Cellular & Molecular Medicine in the Lerner Research Institute, and senior author Takhar Kasumov, PhD, adjunct in Gastroenterology and Hepatology at Cleveland Clinic and fulltime faculty at NEOMED, published a clinical research article in The Journal of Clinical Endocrinology & Metabolism.
The study, “Glycation Reduces the Stability of APOAI and Increases HDL Dysfunction in Diet-Controlled Type 2 Diabetes,” looks at the role of hyperglycemia-induced glycation on ApoA1 kinetics and stability in patients with diet-controlled type 2 diabetes mellitus (T2DM).
The study found that in people with diabetes, glycation of the ApoA1 molecule causes it to degrade three times faster than in a person without diabetes.
“When ApoA1 is destroyed faster, it is not able to perform its main function for reverse cholesterol uptake, which means taking the bad cholesterol and disposing of it,” says study co-author Sangeeta Kashyap, MD. “The bottom line is that it does not just matter how much good cholesterol you have, but how the cholesterol works to protect you. In people with diabetes, good cholesterol does not work normally to protect them from atherosclerotic heart disease.”
From a clinical perspective, Dr. Kashyap says, the data highlights that normal, or even elevated, HDL levels in T2DM does not equate to adequate functionality. Glycation of ApoA1, she says, is a marker for hyperglycemia-induced HDL dysfunction that blunts the anti-atherogenic and anti-oxidant functions of HDL.
Advertisement
These findings were reached using 2H20-metabolic labeling – a novel heavy water-based non-radioactive technique that looks at the kinetics of HDL by measuring the production and destruction of ApoA1. They study also found ApoA1 instability is related to early glycation of lysine on ApoA1 and associated with glycated hemoglobin (HbA1c) levels – a measure of long-term hyperglycemia.
“The important clinical piece for practitioners is that even if you see normal or high HDL levels, don’t think it’s working in their favor,” Dr. Kashyap says. “If they have high blood sugar, that’s a marker that HDL is not working to protect them from the atherogenic process.
Dr. Kashyap says lowering glucose levels could be a way to restore HDL functionality in people with diabetes. While previous articles theorized that high triglycerides were related to good cholesterol, this study found that HDL functionality is related to ambient glucose levels.
Next steps involve looking at various glucose lowering interventions, including the effects of the diabetic drug Metformin and insulin, to lower glucose levels.
“We’re interested in looking at specific interventions to lower glucose levels to see if these defects are reversible. We believe they are,” Dr. Kashyap says. “This is an early glycation process. If we are able to lower blood sugar levels, we can reverse levels of lysine glycation of ApoAI and restore functionality of HDL.”
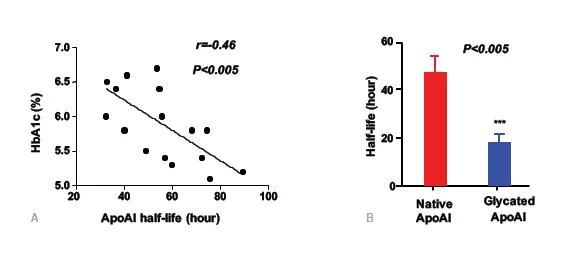
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/3b39f885-4dd7-4a21-b931-27e00311c21d/DrKinset1_jpg)
Apo A1 turnover in Hyperglycemia
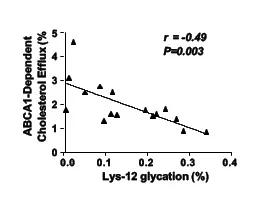
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/65c82667-051e-42b0-9f8f-01d0a40f49c6/DrKinset2_jpg)
Reduced cholesterol efflux of glycated ApoA1
The study was supported by the American Diabetes Association.
Advertisement
Advertisement
Procedure offers potential for less pain and faster recovery in selected patients
Challenges include multimorbidity, polypharmacy, limited mobility and sarcopenia
Data could help inform policy and patient-provider conversations
Pheochromocytoma case underscores the value in considering atypical presentations
Advocacy group underscores need for multidisciplinary expertise
A reconcilable divorce
A review of the latest evidence about purported side effects
High-volume surgery center can make a difference