Shared decision-making tool published in a recent study fills void
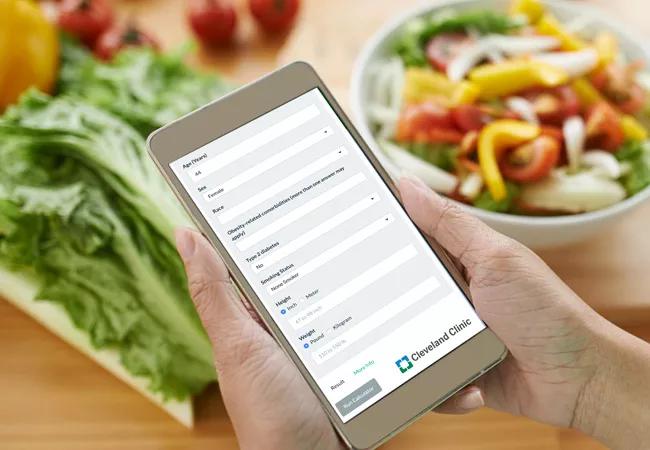
Although there are now five FDA-approved anti-obesity medications (AOM) on the market, a lack of head-to-head trials comparing their effectiveness can make prescribing difficult. A new study from Cleveland Clinic addresses this paucity of real-world prescribing data with the creation of an anticipated weight loss calculator.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
“Physicians may be reluctant to prescribe AOMs for a number of reasons,” states Bartolome Burguera, MD, PhD, Chairman of Cleveland Clinic’s Endocrinology & Metabolism Institute and senior author on the paper. “Some may be uncertain which medication will work best for a particular patient or feel that there is not enough long-term experience with newer medications. Others physicians may not view obesity as a disease that warrants medical intervention, or may feel like they lack the time or training to treat obesity.”
The retrospective study was designed to evaluate weight loss in more than 3,000 adults with BMI ≥30 kg/m2 and no related comorbidities, or ≥27 kg/m2 and at least one obesity-related comorbidity, such as hypertension, type 2 diabetes (T2D), cardiovascular disease or hyperlipidemia. Weight loss data was included for patients who received a 12-week prescription for an FDA-approved AOM along with lifestyle and behavioral interventions. Medications evaluated in this study included phentermine hydrochloride, phentermine-topiramate, bupropion-naltrexone and lorcaserin.
“We chose a 12-week interval to conform with the FDA’s ‘stopping rules,’ which call for a re-evaluation of weight loss medications in those patients who do not reach the 3-5% weight loss threshold within 12-16 weeks of use,” Dr. Burguera explains.
Patients in the study lost an average of 3.45% of their body weight from baseline over the 12-week treatment course. Although patients lost weight on all four medications, patients prescribed phentermine hydrochloride (loss of 3.75±5.66% of body weight) or phentermine-topiramate (3.63±5.7%) lost more weight than those taking lorcaserin (2.66±5.03%) or bupropion-naltrexone (1.84±6.69%). There was no statistically significant difference in percentage of weight loss in patients taking phentermine hydrochloride or phentermine-topiramate.
Advertisement
“We note several interesting findings in this study,” says Kevin Pantalone, DO, an endocrinologist and study author. “Patients were observed to have lost less weight in our study when compared to the weight loss that was observed in randomized-controlled trials evaluating the effectiveness and safety of AOM therapy. This may relate more to the design of our study than to the medications themselves, as medication costs were not offset in our study, we included patients with comorbidities, and we did not control for dose titration. In addition, as these medications are not routinely covered by most insurance plans, medication compliance in the real-world setting is often reduced and challenging to achieve.”
Although the magnitude of weight loss was less than other trials, approximately half of the patients lost at least 3% of their baseline body weight. According to Dr. Burguera, even this modest weight loss is enough to improve glycemic control, reduce cardiovascular risks, improve quality of life and keep patients motivated.
Note: Drs. Burguera and Pantalone would like to thank Kelly Shibuya MD for her help conducting this study during her tenure as a medical student at Cleveland Clinic Lerner College of Medicine.
Advertisement
Advertisement

Pheochromocytoma case underscores the value in considering atypical presentations

Advocacy group underscores need for multidisciplinary expertise

A reconcilable divorce

A review of the latest evidence about purported side effects

High-volume surgery center can make a difference

Advancements in equipment and technology drive the use of HCL therapy for pregnant women with T1D

Patients spent less time in the hospital and no tumors were missed

A new study shows that an AI-enabled bundled system of sensors and coaching reduced A1C with fewer medications