Locations:

Reassuring results, but patient education is still important
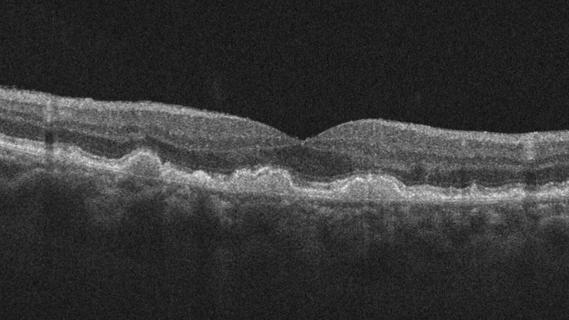
Early data show risk is 73% higher in patients with lupus, 40% higher in patients with rheumatoid arthritis
Transition clinic marks one year of experience

At ACG, Cleveland Clinic gastroenterologist explains benefits of patient-centered IBD medical home and neighborhood
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy

Cleveland Clinic conducts comprehensive review of IBD's widespread effects
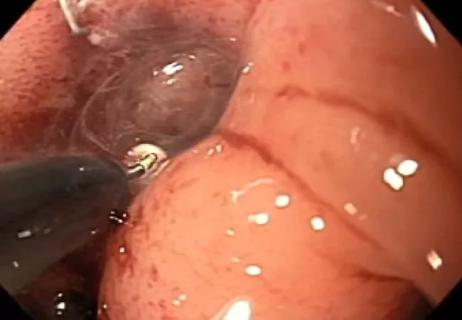
Avoiding complex reoperative abdominal surgery and ostomy
Advertisement
Advertisement