Tag debug info: client: {"assets":{},"datasets":{},"live":{},"projects":{},"users":{},"observable":{"assets":{},"datasets":{},"live":{},"projects":{},"users":{}}} Now: 1770555072481 Cache Key: cqdTagPageBySlug:study fetchCache[cqdTagPageBySlug:study].expirationTime: falsey fetchCache[cqdTagPageBySlug:study]. seconds remaining: falsey All fetchCache expiration times: -- Key: cqdNotFoundPage, seconds remaining: 6182 -- Key: cqdTagPageBySlug:hypertrophic-obstructive-cardiomyopathy-hocm, seconds remaining: -4705 -- Key: cqdPostsByTag:cqd-migrated-tag-21052,1,10, seconds remaining: -4619 -- Key: cqdTagPageBySlug:remote-access, seconds remaining: -3129 -- Key: cqdPostsByTag:cqd-migrated-tag-24304,1,10, seconds remaining: -3075 -- Key: cqdTagPageBySlug:multi-omics, seconds remaining: 519 -- Key: cqdPostsByTag:cqd-migrated-tag-27059,1,10, seconds remaining: 569 -- Key: cqdTagPageBySlug:brain-study, seconds remaining: 1640 -- Key: cqdPostsByTag:cqd-migrated-tag-24300,1,10, seconds remaining: 1693 -- Key: cqdTagPageBySlug:interdigitation, seconds remaining: 2421 -- Key: cqdPostsByTag:cqd-migrated-tag-20954,1,10, seconds remaining: 2471 -- Key: cqdTagPageBySlug:voiding-cystourethrogram-vcug, seconds remaining: 3447 -- Key: cqdPostsByTag:cqd-migrated-tag-885,1,10, seconds remaining: 3495 -- Key: cqdTagPageBySlug:absorb, seconds remaining: 4700 -- Key: cqdPostsByTag:cqd-migrated-tag-37,1,10, seconds remaining: 4768 -- Key: cqdTagPageBySlug:instability, seconds remaining: 6182 -- Key: cqdPostsByTag:cqd-migrated-tag-25293,1,10, seconds remaining: 6229 -- Key: cqdTagPageBySlug:wellness-programs, seconds remaining: 8102 -- Key: cqdPostsByTag:cqd-migrated-tag-4979,1,10, seconds remaining: 8147 -- Key: cqdTagPageBySlug:hyperacusis, seconds remaining: 8974 -- Key: cqdPostsByTag:cqd-migrated-tag-1775,1,10, seconds remaining: 9026 -- Key: cqdTagPageBySlug:pocus, seconds remaining: 9253 -- Key: cqdPostsByTag:cqd-migrated-tag-25622,1,10, seconds remaining: 9311 -- Key: cqdTagPageBySlug:matthew-eagleton, seconds remaining: 9610 -- Key: cqdPostsByTag:cqd-migrated-tag-818,1,10, seconds remaining: 9662 conditions: -- false, -- NA, -- NA, -- NA -- false Cache miss for key cqdTagPageBySlug:study - retrieving from Sanity CCCache.dataFetchCount: 15805 Cache cleanup seconds remaining: 15295
Advertisement
Advertisement
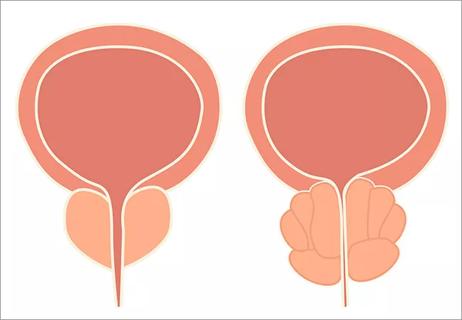
Study of over 5,000 patients shows type of surgery matters too
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
Rendered: Sun Feb 08 2026 12:51:12 GMT+0000 (Coordinated Universal Time)
9500 Euclid Avenue, Cleveland, Ohio 44195 |
800.223.2273 | ©
2026 Cleveland Clinic. All Rights Reserved.