Gene expression analysis uncovers clues
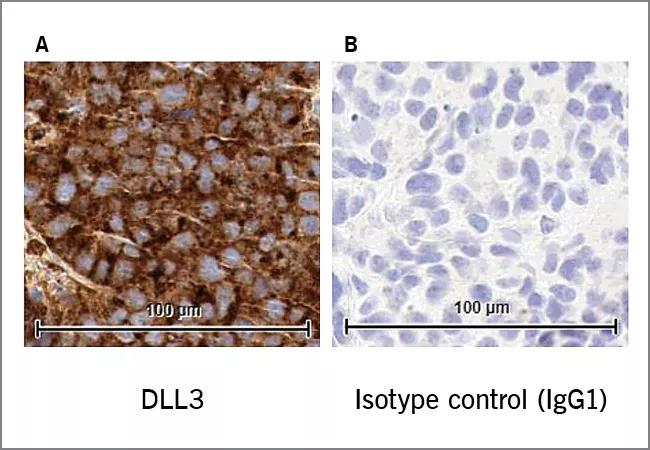
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/ee314a03-765e-4ba9-8411-48a1a390f9ea/19-CNR-574_650x450_jpg)
19-CNR-574_650x450
A recent study published in Clinical Cancer Research has shed light on the biology of small cell bladder cancer (SCBC), a rare, often deadly and poorly understood form of bladder cancer. SCBC is difficult to diagnose and treat as it often progresses quickly, and patients tend not to respond well to current treatment methods drawn from urothelial and small cell lung cancer protocols.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
The biology of SCBC is poorly understood, but its clinical behavior shares similarities with small cell and neuroendocrine tumors of other primary sites, such as small cell lung cancer. Furthermore, modern genetic studies in bladder cancer have largely excluded this rare but important variant, creating a need to better define the molecular biology of SCBC. The investigators used an institutional cohort to test the hypothesis that SCBC tumors express a unique molecular signature that may distinguish SCBC from other, similar diseases, e.g., small cell carcinoma of the lung, and that more importantly could point toward better biomarkers and therapies.
Using pathology records, Omar Mian, MD, PhD, Radiation Oncology and Translational Hematology and Oncology Research, and his colleagues identified 63 patients with SCBC treated at Cleveland Clinic between 1993 and 2016. Data included fixed and archived tissue from about 40 tumors. Additionally, six cases had matched pairs of tumor and adjacent normal tissue, and one case included a sample from a metastatic lesion. They presented initial data at ASTRO 2017 and published a full report in January 2019.
“That’s powerful,” Dr. Mian says, “because we were able to make comparisons in the same individual between their normal bladder gene expression and the tumor-related activation and suppression of genes.”
The researchers found that not all of the 63 tumors were purely SCBC histologically. Some contained a mix of SCBC cells and other bladder cancer cells such as urothelial cancer cells. “It varied anywhere from a small component of the bladder cancer to the entire tumor being small cell,” says Dr. Mian.
Advertisement
Dr. Mian and colleagues also performed differential gene expression analysis on the six matched pairs of tumor and adjacent normal tissue, finding a total of 583 differentially expressed genes. The top differentially expressed genes were primarily involved in cellular functions related to biological regulation of growth, protein binding and metabolism.
The research team analyzed differentially expressed genes based on small cell component of the tumor and performed correlations with immunostaining for known pathologic biomarkers associated with small cell cancers. This analysis generated a novel gene signature for SCBC, which the research team believes will allow distinction of SCBC from other bladder cancer variants based on molecular profiling.
Validation of this gene signature in larger cohorts at Cleveland Clinic and through collaborating institutions is ongoing. If validated, a gene expression signature may, for the first time, provide a reliable molecular pattern for distinguishing this rare subtype of bladder cancer with potential utility in early detection and selection of appropriate patients for tailored treatments.
The molecular analysis of SCBC led to the discovery of several important prognostic and predictive factors. For instance, tumors with DLL3 (Delta Like Canonical Notch Ligand 3) protein expression of greater than 10 percent were associated with shorter OS and PFS from diagnosis and shorter OS from surgery. To test whether DLL3 might serve as a therapeutic target, a DLL3-targeting antibody-drug conjugate was tested in a patient-derived xenograft model and was found to be highly effective in models of SCBC derived from otherwise highly resistant tumors. This exciting finding could lead to a new generation of promising clinical trials in SCBC.
Advertisement
This study is the first to assess prognostic value in proteins associated with neuroendocrine differentiation and is one of the largest of gene expression profiling in SCBC.
More information at mianlab.org
Image: Originally published as Figure 5 in above-referenced article. DLL3 IHC of FFPE sample of a SCBC patient-derived xenograft (PDX) model (A) and isotype control (IgG1) of same FFPE sample showing lack of staining (B).
Advertisement
Advertisement
New guidelines empower clinicians with practical diagnostic framework
Pivotal Studies Guide Treatment Decisions in Muscle-Invasive Bladder Cancer
Reflections from an oncology provider and communications educator on new ASCO Guidelines on Patient-Clinician Communication
Phase 1 trial outcomes offer encouraging news for developing targeted therapy for solid tumors
Cleveland Clinic to administer first-of-its-kind T-cell therapy
Heavily pretreated patients experience improved progression-free survival and quality of life with CD20xCD3 therapy
Innovative procedure reduces scarring, recovery time and nipple sensation
Best practices for supporting patients with honesty and compassion