Series of five patients successfully treated with ‘ventricular switch’
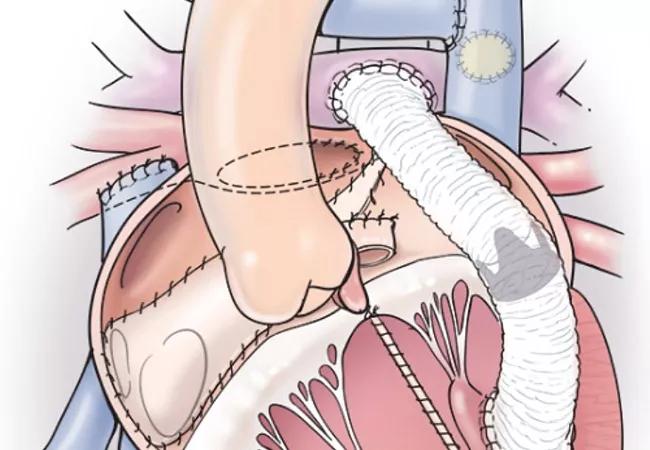
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/fece44e2-fa2a-494a-8c6e-09296750f797/20-HVI-1872559-Biventricular-Conversion-in-Unseptable-Hearts-CQD-Hero_jpg)
biventricular conversion for unseptatable hearts
Five patients with anatomically complex congenital heart disease that would typically be addressed with single-ventricle palliation successfully underwent a biventricular conversion approach in which the morphologic left ventricle was used as the subpulmonary ventricle. The procedure, which resulted in immediate resolution of hypoxia and improved symptoms and functional health, was reported along with the case series by Cleveland Clinic clinicians at the virtual meeting of the American College of Cardiology (ACC) in late March.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
“What we named the ‘ventricular switch’ procedure allows a two-ventricle circulation to be created in patients traditionally considered ‘unseptatable’ due to the inability to route the left ventricle to the aorta,” says Hani Najm, MD, Chair of Pediatric and Congenital Heart Surgery at Cleveland Clinic, who led the team performing the surgeries. “This complex novel approach is fully reproducible and made possible by extensive preparation using advanced imaging and 3D-printed anatomic models.”
Complex systemic and pulmonary venous anatomy, common atrioventricular canal defects, abnormal ventricular or atrial arrangement, and conotruncal anomalies are traditionally regarded as being unseptatable, often with a grim prognosis. Single-ventricle palliation offers poor long-term survival and few options for rescue. But attempting a biventricular repair in patients with these types of anatomy is risky, with substantial perioperative morbidity and mortality as well as uncertain long-term quality-of-life outcomes and survival benefits.
Advanced imaging technology combined with 3D printing is making surgeries possible that were once considered too complex to safely attempt, according to Dr. Najm. In the Cleveland Clinic case series presented at the virtual ACC meeting, MRI or CT was used to construct 3D replicas of the heart that differentiate cardiac structures by color. The technology enabled the surgical team to determine whether biventricular conversion was feasible and to define in detail the steps needed for septation.
Advertisement
“Unlike traditional images, 3D-printed models provide a way to facilitate iterative refinement of different surgical approaches,” Dr. Najm explains. “The models also enable us to share our experience with other surgeons and centers.”
Five patients — ages 11 months, 5 years, 9 years, 31 years and 46 years — were operated on between 2016 and 2019. All had complex heterotaxia that was considered unseptatable, and all were severely cyanotic and had extreme functional limitations. Among four patients, a total of 12 previous cardiac surgeries had been performed elsewhere in anticipation of univentricular repair.
All patients required complex atrial septation, and three underwent ventricular septation (precluded by fixed pulmonary resistance in the other two). The ventricular switch refers to diverting the pulmonary venous return to the morphologic right ventricle as the systemic ventricle. A two-ventricle repair (1.5-ventricle repair in one patient) was then performed, informed by the potential capacity of the left ventricle based on MRI-indexed volumes.
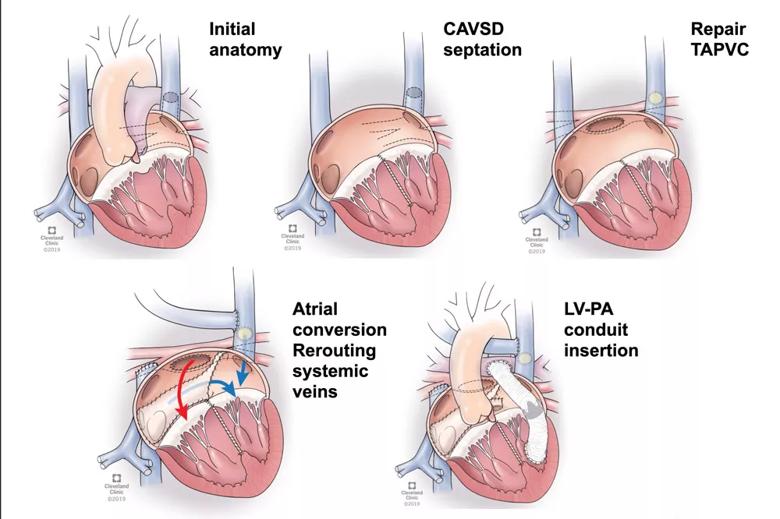
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/47911323-6e14-4478-b416-4e6b679a887d/20-HVI-1872559-Biventricular-Conversion-in-Unseptable-Hearts-CQD-Inset_jpg)
Figure. Illustrations showing key stages of the operative approach. CAVSD = complete atrioventricular septal defect; TAPVC = total anomalous pulmonary venous connection; LV-PA = left ventricle to pulmonary artery.
Median hospital stay was 13 days, with a range of 10 to 60 days. All patients had functional status improvement and significantly increased mean systemic oxygen saturation (from 79% ± 7% at baseline to 95% ± 5% postoperatively; P = 0.003).
Advertisement
All patients were alive at the time of the report, with a median follow-up of 7 months (range, 1 month to 2.7 years).
Dr. Najm and colleagues note that because the report is limited to only five patients at a single center with short follow-up thus far, it does not carry the weight of a prospective study comparing the strategy to univentricular palliation in similar patients. They add, however, that the following physiological considerations support the approach:
The immediate improvement observed in patients postoperatively also supports the procedure’s value and feasibility.
Dr. Najm offers the following general advice regarding this complex surgery:
Advertisement
Each case in the series was unique and required an individual approach, Dr. Najm emphasizes. Although patient age varied widely, he believes that performing the operation as early as possible may lead to the best outcomes. He adds that atrioventricular regurgitation — even in the systemic atrioventricular valve — should not preclude having the procedure, as the valve can be replaced.
Although all five patients had heterotaxia, he suggests that the procedure’s techniques may be relevant to patients with other complex configurations, such as abnormal atrial and ventricular arrangements (e.g., criss-cross heart and ventricular inversion), double-outlet right ventricle with remote ventricular septal defect, and Shone’s variants or other forms of a hypoplastic left heart complex.
“Thanks to advanced technology allowing detailed planning, this approach is extremely versatile,” Dr. Najm concludes.
Advertisement
Advertisement
Cleveland Clinic reports first U.S. series focused on use in this challenging setting
Large series confirms early and long-term survival advantages over partial pericardial resection
AVANT GUARD trial extends first-line role for ablation beyond paroxysmal atrial fibrillation
Maintain a high index of clinical suspicion and consider the underlying etiology
Protocol adoption at Cleveland Clinic sharply raised share of transferred patients getting timely PCI
Intracardiac echo mapping of para-septal fat pads provides fast, accurate and radiation-free targeting
Join us in Cleveland July 17 for a practical, first-of-kind course
Robust signal from observational study raises prospect of a long-sought medical therapy