Cleveland Clinic and Case Western researchers on the forefront
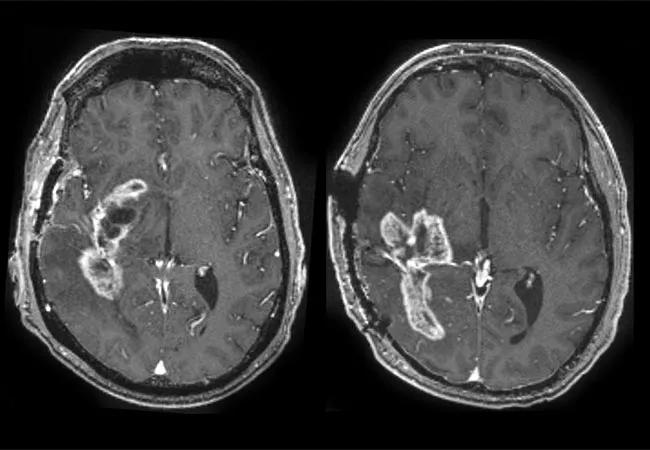
Scientists and clinicians from Cleveland Clinic and Case Western Reserve University recently received a $1.15 million grant from the National Cancer Institute (NCI)’s Informatics Technology in Cancer Research (ITCR) program to develop artificial intelligence (AI) tools that assist in the differentiation of tumor recurrence versus scar tissue on postoperative MRI scans in patients with certain colorectal and brain cancers.
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
Manmeet Ahluwalia, MD, MBA, the Dean and Diane Miller Family Endowed Chair in Neuro-Oncology in Cleveland Clinic’s Rose Ella Burkhardt Brain Tumor and Neuro-Oncology Center, is one of the grant recipients. “In patients with glioblastoma (GBM) treated with surgical resection and radiotherapy, the brain’s reaction to radiation can mimic the appearance of tumor progression on MRI. So when we perform the requisite post-treatment surveillance imaging, we need a way to distinguish between the two so that we don’t perform unnecessary procedures or miss tumor growth,” he says.
Difficulty distinguishing between scar tissue and tumor recurrence on MRI can also be an issue for patients with colorectal cancer. A recurring tumor on MRI in these patients can mean a proctectomy and significant reduction in quality of life. A more precise reading of MRI results could spare many patients unnecessary procedures.
The tool under development uses the power of the deep-learning computers at the Case School of Medicine and Engineering Center for Computational Imaging and Personal Diagnostics (CCIPD).
“Our recent research applies feature engineering and machine learning techniques to conventional MRI to quantify subtle distinguishing features not normally appreciated by the unaided eye. Such features, which we call radiomics, can include details of lesion shape, relation of enhancing margins to adjacent edematous margins, eccentricity of the lesion, texture of the lesion, as well as domain-inspired features relating to the mass effect deformation, and the organization of pixels or gradients within the lesion,” notes Pallavi Tiwari, PhD, Case Western Reserve School of Medicine faculty and lead researcher on the project.
Computerized assessment’s three-dimensional approach is superior to the two-dimensional assessment of the human eye. The grant will allow continued development of machine learning techniques that use this three-dimensional assessment to develop an algorithm by assessing a large number of examples and then learning distinguishing features.
“This approach will also allow us to envision the entire tumor environment,” says Dr. Ahluwalia. “Normally, we just have a tissue sample from a biopsy, which can be inaccurate depending on the location from which it was obtained.”
Part of the grant calls for the research team to disseminate the tool to other scientists, as radiomics is not widely understood in research and clinical communities. As radiomics and machine learning algorithms begin to make substantial contributions to medical care, the need for widespread understanding and use grows. “Our algorithms are only as good as the training we give them,” notes Dr. Ahluwalia, “so the more research we do, the more precise understanding we can have of the disease in question, which leads to better decisions for our patients.”
This is the third concurrent NCI/ITCR grant at the CCIPD focusing on AI tools for cancer diagnosis and prognosis. For more information about machine learning advances in brain cancer, read this article written by members of the grant team.

Timing and type of side effects differ greatly from chemotherapy

Dedicated multidisciplinary teams support 84 ultra-rare cancers

Sessions explore treatment advances and multidisciplinary care
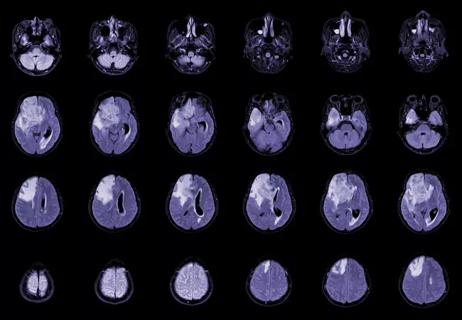
New research from Cleveland Clinic helps explain why these tumors are so refractory to treatment, and suggests new therapeutic avenues
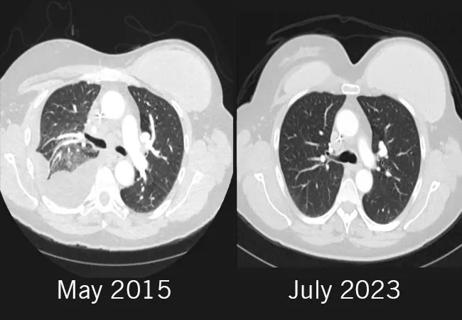
Combination of olaparib and carboplatin results in complete durable response for a patient with BRCA2 and “BRCAness” mutations
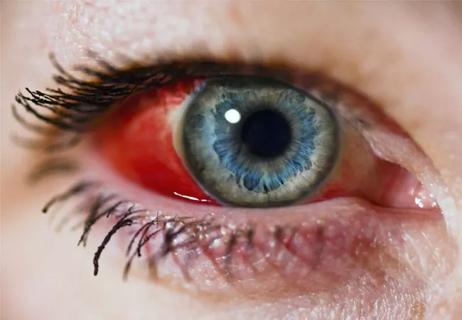
Early communication between oncologists and ophthalmologist warranted

Case-based course delves into latest treatment approaches

Long-term relationship building and engagement key to gaining community trust