New reports focus on neuroimaging biomarkers and features of atypical presentations
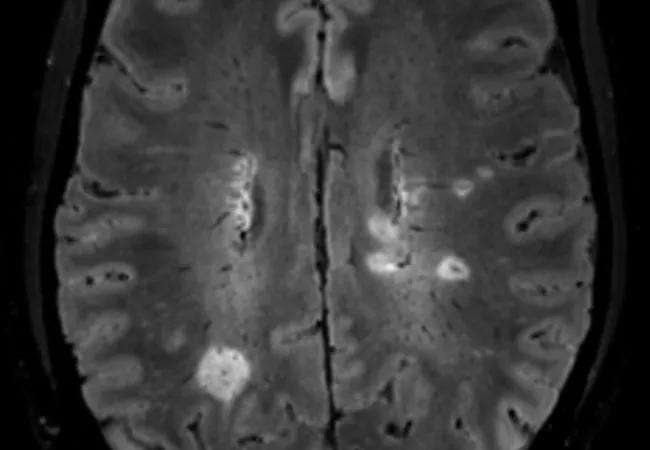
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/21f52ebc-fdd2-41e1-bf94-86dc70895431/20-NEU-1845659-CQD-Ontaneda_Grant-to-study-central-vein-sign-in-MS_650x450_Hero_jpg)
central vein sign on a brain MRI
Detecting both a central vein sign (CVS) and a paramagnetic rim lesion (PRL) on a single MRI scan has good sensitivity and specificity for multiple sclerosis (MS), making this combination of features a promising candidate for improving diagnosis of this often-misdiagnosed condition. A noninvasive point-in-time method for diagnosing MS has been a holy grail in the field, as diagnosis currently relies on establishing dissemination of the disease in time and space and occasionally warrants invasive testing such as lumbar puncture or continued clinical monitoring due to diagnostic uncertainty.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
Evidence for this potential advance was presented as a poster at the recent Americas Committee for Treatment and Research in Multiple Sclerosis (ACTRIMS) Forum 2024 by a team of researchers from multiple institutions in the U.S. and Canada, co-led by Cleveland Clinic neurologist Daniel Ontaneda, MD, PhD. Another poster presentation led by this group focused on characterizing typical versus atypical clinical presentations of MS, also with the goal of improving diagnosis.
“Misdiagnosis occurs in almost 20% of patients who present with MS, at times exposing individuals to potentially risky treatments,” Dr. Ontaneda says. “Both of these avenues of research show great promise for improving accuracy.”
Both posters were related to the CAVS-MS trial (Central Vein Sign: a Diagnostic Biomarker in Multiple Sclerosis) — either its completed pilot study or the ongoing primary CAVS-MS investigation (NCT04495556).
The primary CAVS-MS trial (detailed in a prior Consult QD post) is a prospective biomarker trial investigating the use of CVS as a specific diagnostic marker for MS in a mixed population of patients with typical and atypical presentations. More than 400 patients presenting to a North American MS center for diagnosis are being followed for 24 months, during which they undergo clinical and neuroimaging assessment with FLAIR* MRI, the special imaging method used to detect central veins. Subjects were enrolled prospectively without knowledge of their diagnosis; a subsequent diagnosis of MS is based on the 2017 McDonald criteria. Patient follow-up is expected to be complete in late 2024. Dr. Ontaneda is co-principal investigator of this multicenter study.
Advertisement
The CAVS-MS pilot study, designed primarily to optimize and harmonize FLAIR* MRI among 10 trial sites in North America, was reported on in a prior Consult QD post.
This investigation, based on the CAVS-MS pilot study, used a cohort of 37 adults with MS and 41 with an alternative diagnosis to assess the diagnostic performance of the combined presence of neuroimaging signs, an area of investigation that has not been well studied.
Prevalence of CVS+ lesions (seen on post-gadolinium FLAIR* images) and PRL+ lesions (derived from high-resolution T2*-weighted echo-planar imaging) differed markedly at baseline between the two groups: the combined presence of at least one CVS+ lesion and at least one PRL+ lesion occurred in 32 MS participants (86%) as opposed to three participants (7%) with an alternative diagnosis. Diagnostic accuracy for MS was 90%, with a sensitivity of 0.86 (95% CI, 0.71-0.95) and specificity of 0.93 (95% CI, 0.80-0.98).
“The diagnostic specificity of the presence of both biomarkers on the same scan was comparable to the detection of oligoclonal bands in cerebrospinal fluid, and sensitivity was actually better,” says Karlo Toljan, MD, a Cleveland Clinic clinical research fellow in neuroimmunology and first author of the poster. “Finding a neuroimaging approach with such high accuracy is very encouraging for potentially gaining a valuable new method for establishing a correct diagnosis of MS.”
He notes that the biomarkers can be demonstrated on standard 3T MRI scans, so the tool should be widely available to neuroradiologists in MS care.
Advertisement
“Basing a diagnosis purely on neuroimaging biomarkers enables clinicians to make a diagnosis regardless of symptoms and when the patient presents,” Dr. Ontaneda adds. “This research, demonstrating very specific biomarkers of MS, is an important step in that direction.”
Many patients who present for evaluation of MS have symptoms typical of the disease but do not have typical clinical attacks. These individuals with atypical presentations have no pathway to a diagnosis of MS, as the criteria require a typical attack or true neurological progression for a diagnosis to be made. Clinicians currently need to wait for these patients to develop an attack or progression before they can be diagnosed, and identifying which of these individuals are at highest risk of MS is an important area of research.
This investigation used a cohort of 420 patients from the ongoing CAVS-MS biomarker trial who were determined by a team of five neurologists to either have a typical MS attack (n = 209) or not (n = 211).
The most common presenting symptoms among patients with typical MS attacks included the following:
Among patients without typical attacks, 141 had atypical onset of symptoms, with the most common being:
In this group with atypical symptom onset, 21% met MRI criteria for dissemination in space (DIS) and 5% met both MRI DIS and dissemination in time (DIT) criteria.
Advertisement
“It’s interesting to note that the most common presentations for both typical and atypical MS presentations were sensory symptoms,” Dr. Ontaneda says. “The designation of whether sensory symptoms are compatible with a true clinical attack is somewhat arbitrary, and this can be the difference between being diagnosed with MS or not, which is problematic. The significant percentage of the atypical group who radiologically fulfill DIS and/or DIT criteria attests to this. I suspect that those patients with suggestive radiological findings actually have MS. This study highlights the importance of neurologists’ judgment in maneuvering through the diagnostic criteria to try to reach an accurate diagnosis.”
Dr. Ontaneda emphasizes the importance of long-term data for determining the significance of neuroimaging biomarkers, which will be available from the CAVS-MS trial. “The research findings presented in these posters support the possibility of moving away from a diagnosis of MS requiring clinical characterizations to greater reliance on objective and specific biomarkers,” he concludes. “I expect we will see some changes in that direction in the next iteration of the McDonald criteria.”
Advertisement
Advertisement
Mixed results from phase 2 CALLIPER trial of novel dual-action compound
A co-author of the new recommendations shares the updates you need to know
Rebound risk is shaped by patient characteristics and mechanism of action of current DMT
First-of-kind prediction model demonstrates high consistency across internal and external validation
Real-world study also finds no significant rise in ocrelizumab-related risk with advanced age
Machine learning study associates discrete neuropsychological testing profiles with neurodegeneration
This MRI marker of inflammation can help differentiate MS from mimics early in the disease
Focuses include real-world research, expanding access and more