Ultrahigh field strength increases detection sensitivity and temporal resolution
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
Patients suffering from intractable focal epilepsy potentially can be cured if the epileptogenic zone is identified and surgically resected. While current techniques such as scalp and intracranial EEG, structural MRI, MEG and PET are often helpful in identifying this zone, many patients remain untreated due to inadequacy of these techniques, a complicated disease presentation or both. For this reason, there is a continued need to develop new techniques to help identify the epileptogenic zone.
One promising technique is simultaneous EEG-fMRI, wherein sufficiently large epileptic spikes cause an associated change in local blood flow, which then can be visualized using a blood-oxygen-level-dependent (BOLD)-sensitive sequence during functional MRI (fMRI). This technique was first described nearly 20 years ago and classically requires EEG recordings to be obtained simultaneously during an MRI scan. These recordings then can be used to identify the time points of any spikes occurring during the MRI, which allows postprocessing methods to visualize the three-dimensional location of the epileptogenic spike. The advantage of adding MRI to identify the location is that its spatial accuracy is far superior to that of scalp EEG recordings alone.
Nevertheless, there are several drawbacks to this promising method:
Advertisement
At Cleveland Clinic, we have recently explored ways to address these difficulties by extending the earlier technique to ultrahigh-field MRI (7 tesla [7T], versus standard 1.5T or 3T), which first required developing a new head coil for safe operation and extensive testing. Using 7T MRI provides several advantages:
We have tested the method using a simple model of a spike and its associated network: Volunteers have been scanned while in the resting state except for performing a single volitional tap of the index finger in response to an auditory stimulus. This method can easily detect the known BOLD activation maps for the associated auditory and motor networks. Thus we hypothesize that the method should be able to detect any spike whose BOLD response is at the level of a single finger tap or greater.
After confirming the model with tests, we have now applied this method to eight patients with epilepsy. Example findings from two patients are presented in Figures 1 and 2, both of which show close correspondence of EEG-fMRI localization to other localization modalities, as detailed in the captions. These early data provide evidence to support future studies to thoroughly investigate this noninvasive method.
Advertisement
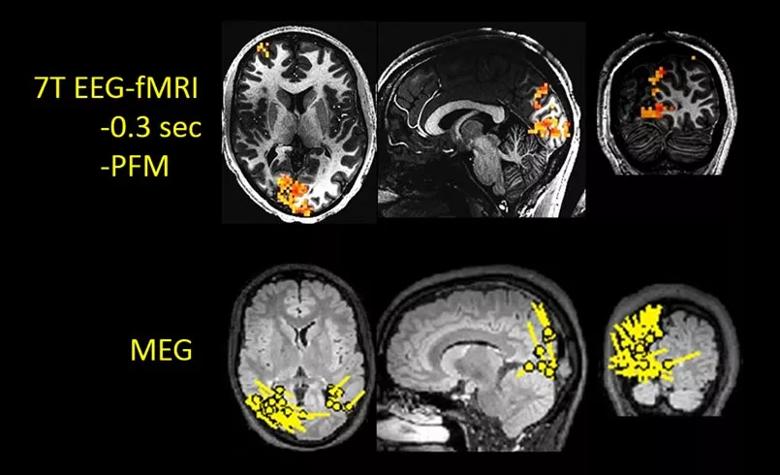
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/a12fdf26-e927-4d5d-aaeb-ec1714f07476/18-NEU-5806-EEG-fMRI-MEG-epilepsy-Fig1_jpg)
Figure 1. Example of 7T EEG-fMRI applied to an epilepsy patient with a known occipital lobe focus, as displayed by the single-dipole models from a MEG study in the bottom row (performed before 7T EEG-fMRI). The top row shows three planes through the right occipital lobes, where the red overlaying the gray anatomy shows a strong focus of BOLD activation. Note the close correspondence of the BOLD location to that shown on MEG. PFM = paradigm free mapping.
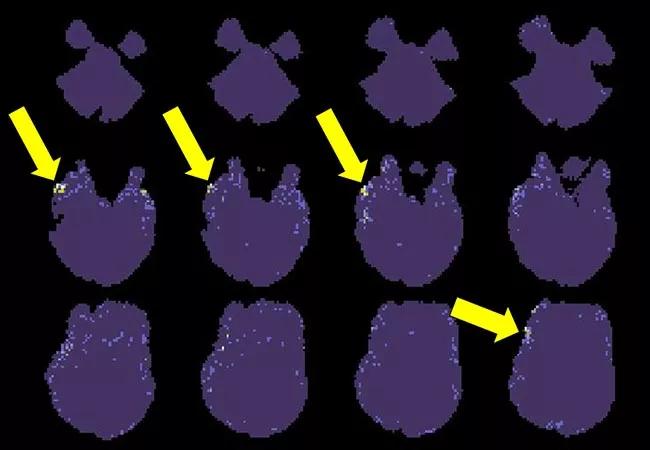
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/74b7433b-b9a8-467c-b945-0b07e911e5fb/18-NEU-5806-Jones-epileptogenicity-EEG-fMRI-650x450_jpg)
Figure 2. Findings of 7T EEG-fMRI recently applied to a 24-year-old right-handed man with a three-year history of epilepsy now treated with multiple medications. While the structural MRI is negative, both PET and video-EEG indicated focal epilepsy likely arising from the right temporofrontal regions. The displayed EEG-fMRI maps were obtained without timing information from EEG, as computed from four BOLD spikes. Corresponding to the other findings, these maps indicate a small network of activation (yellow arrows) in the right frontotemporal region.
Any noninvasive MRI technique that can guide neurosurgeons to resect the region of brain tissue causing epilepsy holds enormous potential benefit. We are addressing the need for such a technique by extending traditional EEG-fMRI methods to 7T MRI, which markedly increases detection sensitivity and temporal resolution of epileptogenic spikes, and can be performed in a data-driven manner.
Acknowledgments. The author acknowledges contributions to this work from César Caballero-Gaudes (Basque Center of Cognition, Brain and Language, San Sabastian, Spain); from Anna Crawford; Mark Lowe, PhD; Sehong Oh, PhD; and Wanyong Shin, PhD (Imaging Institute, Cleveland Clinic); and from Balu Krishnan, PhD, and Imad Najm, MD (Neurological Institute, Cleveland Clinic).
Advertisement
Dr. Jones is a staff neuroradiologist and Vice Chair for Research and Academic Affairs in Cleveland Clinic’s Imaging Institute.
Advertisement
Advertisement

Aim is for use with clinician oversight to make screening safer and more efficient

Rapid innovation is shaping the deep brain stimulation landscape

Study shows short-term behavioral training can yield objective and subjective gains

How we’re efficiently educating patients and care partners about treatment goals, logistics, risks and benefits

An expert’s take on evolving challenges, treatments and responsibilities through early adulthood

Comorbidities and medical complexity underlie far more deaths than SUDEP does

Novel Cleveland Clinic project is fueled by a $1 million NIH grant

Tool helps patients understand when to ask for help