EBUS-TBNA found safe and effective
In a study published in the June 2018 issue of the Journal of Bronchology and Interventional Pulmonology, Cleveland Clinic’s team of researchers found endobronchial ultrasound-guided transbronchial needle aspiration (EBUS-TBNA) to be safe and effective in the diagnosis of EBUS-visible intrapulmonary lesions.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
Francisco A. Almeida, MD, MS, interventional pulmonologist in Cleveland Clinic’s Department of Pulmonary Medicine and Jordan Reynolds, MD, anatomic pathologist in Cleveland Clinic’s Department of Laboratory Medicine and Pathology, both co-authors of the study, comment on the significance of their latest findings.
“While the EBUS-TBNA procedure is well established in the mediastinal staging of non-small cell lung cancer (NSCLC), our study is the most comprehensive to show the diagnostic value of EBUS-TBNA for EBUS-visible pulmonary lesions and one of the largest sample series to date,” says Dr. Almeida. “These findings can be used to make important decisions in the evaluation of often difficult-to-sample pulmonary lesions and avoid the need for additional diagnostic procedures, especially in lung cancer.”
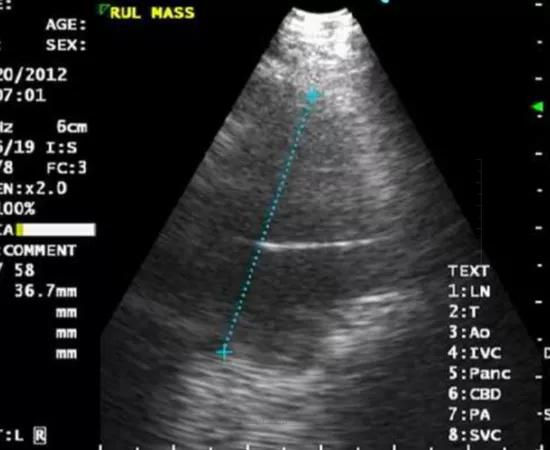
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/697f8f0d-f4fa-4607-8b62-dc5d616d43bb/Case-1-EBUS-image-1_550x450_jpg)
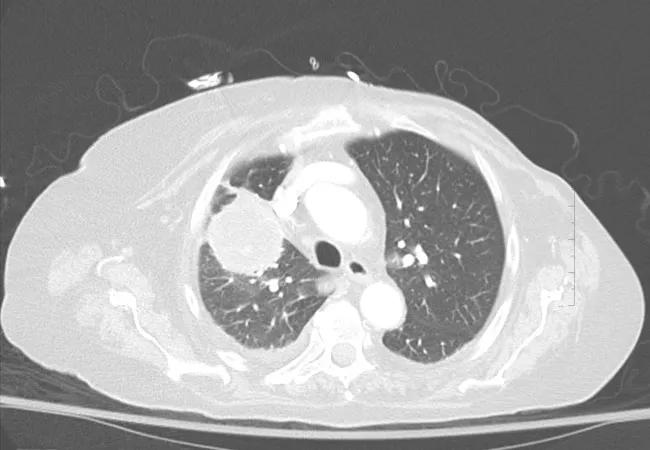
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/290c7a32-c7a1-45e0-b9bb-2fe133a600ac/Case-1-CT-image2_650x450_jpg)
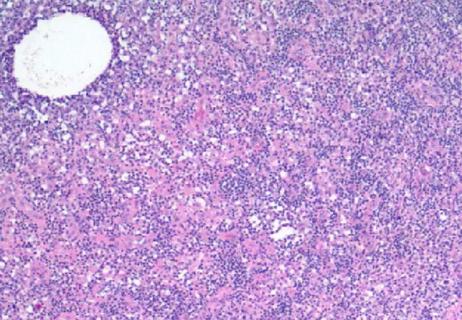
CT showing right upper lobe mass. Sampled with EBUS-TBNA through the trachea. Rapid onsite cytology demonstrated both necrosis and acute inflammation. EBUS-TBNA samples were sent for cultures which demonstrated Nocardia cyriacigeorgica complex.
The study was designed as a retrospective analysis of 108 EBUS-TBNA procedures in 105 patients with EBUS-visible intrapulmonary lesions who underwent the procedure at Cleveland Clinic between January 2010 and March 2015. EBUS-visible intrapulmonary lesions were defined as lesions not visible by white-light bronchoscopy (WLB), but detectable using convex probe endobronchial ultrasound-guided transbronchial needle aspiration (CP-EBUS). Patients with endobronchial lesions that were visible by WLB were excluded from the study.
Advertisement
Among 108 analyzed procedures, EBUS-TBNA returned a positive finding from an EBUS-visible intrapulmonary lesion in 87 percent of cases. The procedure was useful in establishing the following diagnoses: lung cancer (67.6 percent), lung metastases (9.2 percent), infection (4.6 percent), sarcoma/spindle cell sarcoma or neoplasm (2.8 percent), as well as lymphoma, hamartoma and unspecified malignancy (one case each, < 1 percent). EBUS-TBNA did not yield a diagnosis in 13 percent (N = 14) of the cases; of those 14 cases, nine turned out to have benign disease confirmed by additional testing or follow-up imaging, while the remaining five were diagnosed with a malignant condition.
“The sensitivity of EBUS-TBNA to differentiate malignancy from benign disease is high at 94.7 percent,” Dr. Almeida says. “Furthermore, the negative predictive value of EBUS-TBNA in our study was 75 percent, which is pretty good given the high prevalence of malignant disease.”
The American Cancer Society (ACS) estimates that approximately 234,030 new cases of lung cancer will be diagnosed in 2018 in the U.S. alone. The need for safe, effective and minimally-invasive techniques for diagnosing intrapulmonary lesions as early as possible in the course of the disease is clear and underscores the relevance of this study’s results.
“Analyzing samples from intrapulmonary lesions is challenging because, aside from surgical samples, we have to work with very little tissue in order to perform all the necessary testing in the era of targeted lung cancer therapy, including immunotherapy,” explains Dr. Reynolds. “Our study demonstrated that EBUS-TBNA is an effective, minimally-invasive procedure suitable for establishing the diagnosis of EBUS-visible pulmonary lesions, as well as for obtaining specimens for subtyping and cancer biomarker testing.”
Advertisement
Only two serious complications were observed in the analyzed group of patients. “The low rate of serious adverse events (< 2 percent) observed in our study makes this procedure attractive from the safety point of view,” says Dr. Almeida. “In fact, the rate of major complications with EBUS-TBNA appears to be lower than that observed with CT-guided biopsy, which is around 6-7 percent for pneumothorax and bleeding requiring chest drainage and blood transfusion, respectively.”
Dr. Almeida is optimistic that these latest findings will lead to more extensive application of EBUS-TBNA as a single technique for concomitant use in lung cancer diagnosis and staging.
“The safety and effectiveness of EBUS-TBNA demonstrated in our study indicates that EBUS-TBNA should be considered as the diagnostic procedure of choice in patients with EBUS-visible intrapulmonary lesions undergoing lung cancer staging,” Dr. Almeida concludes. “This applies to lesions associated with both NSCLC and small-cell lung cancer.”
Advertisement
Advertisement

Screen patients seeking care for chlamydia, gonorrhea
Lingulectomy removes infection when antibiotics fail

Researchers have developed immunoprofiles for an emerging disease with a mortality rate as high as 27%

Findings from one of the first published case series
Don't discount this crucial step
How to spot the rare infection
Not all lung nodules are malignant
An uncommon condition in a 41-year-old man