Test helpful for SLN-negative, stage I to IIA and thin tumors
A 31-gene expression profile (31-GEP) test offers a predictive metric to overlay onto traditional melanoma staging for better stratification of metastatic risk. Brian Gastman, MD, medical and surgical director of Cleveland Clinic’s melanoma and high-risk skin cancer program, investigated the value of the test for predicting outcomes and guiding treatment for patients within three categories of early stage melanoma.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
The 31-GEP test uses biological information from tumor cells to predict risk of metastasis. “Until this test became available, staging and sentinel node biopsy [SLNB] have been the staples for informing treatment, but they are not sufficient,” Dr. Gastman says. Two out of three patients who develop metastatic disease had a negative SLNB upon diagnosis, and 3 percent of stage IA, 9 percent of stage IB and 19 percent of stage IIA tumors (all considered early stage) will likely prove fatal over a five-year period.
“We have needed enhanced tools to more specifically and accurately define risk. The 31-GEP is a leap forward in solving treatment conundrums for low-risk patients, by identifying those most likely to benefit from intensive surveillance and adjuvant therapy.”
Dr. Gastman led a study to determine 31-GEP’s predictive potential for metastasis in patients within low-risk subpopulations — those who have SLN-negative disease, stage I to IIA tumors and thin tumors. Results were published in the Journal of the American Academy of Dermatology.
He and his co-authors evaluated data from 690 pooled cases of patients with SLN-negative, stage I-IIA or thin tumors. They performed Kaplan-Meier and Cox regression analyses on 31-GEP class (1A-2B, low to high), tumor stage, Breslow thickness, ulceration, mitotic rate and survival outcomes.
Endpoints were recurrence-free survival to either a regional or distant metastatic event; distant metastasis-free survival to any metastatic event beyond the regional nodal basin; and melanoma-specific survival from diagnosis to death from melanoma.
Advertisement
All non-recurrent cases had at least five years of follow-up.
Currently, national treatment guidelines do not include intensive surveillance and adjuvant therapy for patients considered at the lowest risk of metastasis. “With tumors < 1 mm, for example, the National Comprehensive Cancer Network (NCCN) recommends considering SLNB if there are adverse features. We wanted to know if GEP could add information regarding their risk, independent of SLNB recommendation or status, and this data strongly suggests it does,” Dr. Gastman says.
For example, with very thin tumors (Breslow thickness of 1 mm or less), long-term survival rates are so high and the number of these tumors so large that NCCN guidelines do not recommend intensive follow-up and imaging for these patients. But the 31-GEP identifies the small percentage among these low-risk patients whose tumors are more likely to metastasize.
Advertisement
“The test also generates some false positives, but the net effect is that a small proportion of additional patients are earmarked for close scrutiny, and many in this subgroup will have been candidates for metastatic disease,” Gastman explains.
“Therapies are more effective when the tumor burden is low, and since the 31-GEP enables us to ferret out the higher-risk among ‘low-risk’ patients, we are primed for identifying these patients and implementing appropriate clinical follow-up and imaging to discover metastases as early as possible.”
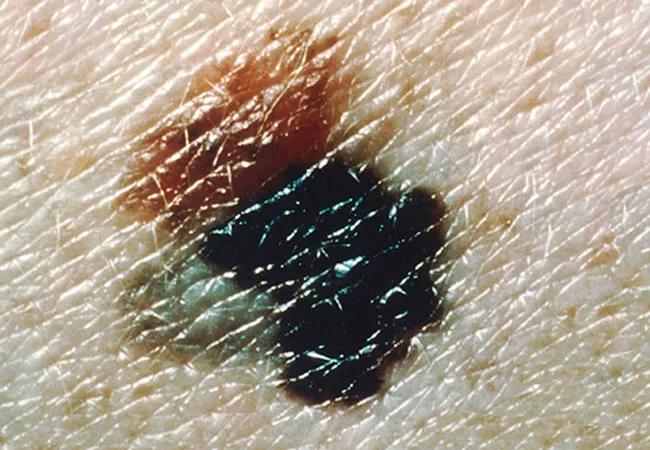
Feature image: Superficial spreading melanoma arising from a dysplastic nevus. The 4-by-8-mm, pink-tan lesion with irregular borders at the upper left (arrow) is a dysplastic nevus. Arising from it is an invasive malignant melanoma, with its characteristic blue-black color, notched border and distorted surface. The gray area at the lower left represents tumor regression. Source: NCI Visuals Online.
Advertisement
Advertisement

First full characterization of kidney microbiome unlocks potential to prevent kidney stones

Researchers identify potential path to retaining chemo sensitivity

Large-scale joint study links elevated TMAO blood levels and chronic kidney disease risk over time

Investigators are developing a deep learning model to predict health outcomes in ICUs.

Preclinical work promises large-scale data with minimal bias to inform development of clinical tests

Cleveland Clinic researchers pursue answers on basic science and clinical fronts

Study suggests sex-specific pathways show potential for sex-specific therapeutic approaches

Cleveland Clinic launches Quantum Innovation Catalyzer Program to help start-up companies access advanced research technology