Endovascular navigation technology clears first preclinical study
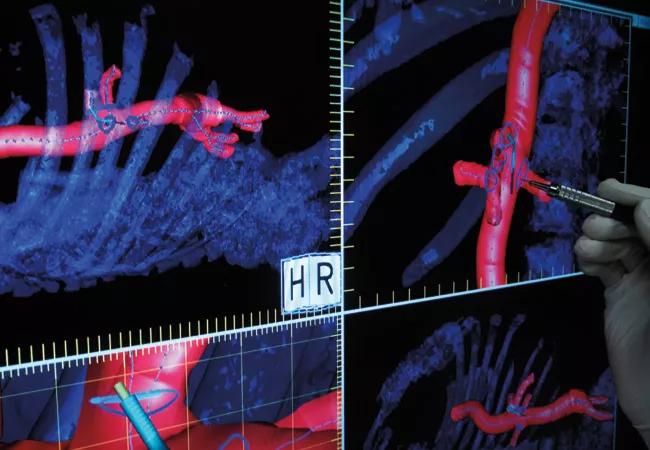
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/7e67ed99-f224-4347-ba85-8d8d56e097db/16-HRT-1407-Centerline-IOPS-650x450_jpg)
Centerline Biomedical IOPS
Endovascular repair of aortic aneurysms and dissections is critically dependent on optimal understanding of the patient’s vasculature. To that end, Cleveland Clinic Innovations spinoff company Centerline Biomedical Inc. is refining development of its Intraoperative Positioning System (IOPS), a novel endovascular navigation technology developed by Cleveland Clinic researchers.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
IOPS extracts the centerlines of the aorta and branch vessels from a patient’s CT and then mathematically constructs a high-quality three-dimensional model of the relevant vasculature, as shown in the image above. Electromagnetic tracking technology allows the model to be used in a GPS-like fashion to guide surgeons during minimally invasive endovascular aortic repairs, enabling less reliance on X-ray fluoroscopy.
The first preclinical in vivo study of IOPS was completed in May 2016 at Cleveland Clinic Lerner Research Institute. It demonstrated the system’s ability to provide non-radiation-based navigation with superior visualization compared with X-ray fluoroscopy, which is limited by its two-dimensional visualization and by the radiation exposure it confers on patients and providers.
“Preclinical evaluation verified the ability to use this navigation system in manipulating through the aorta and its branches in the absence of radiation-inducing fluoroscopy,” says Cleveland Clinic vascular surgeon Matthew Eagleton, MD, who led the study. Noting that catheters and guidewires were easily visualized on the IOPS display, he predicted that the system “will be a game changer in the era of endovascular therapy.”
Centerline Biomedical plans to submit IOPS for FDA review, with a target market entry in the third quarter of 2017.
Advertisement
Advertisement
Large retrospective analysis may prompt prospective studies
How to talk about lifetime risk, treatment goals, Lp(a) testing, statin skepticism and more
A scannable recap of recent volumes and clinical metrics from Cleveland Clinic
Cleveland Clinic reports first U.S. series focused on use in this challenging setting
Large series confirms early and long-term survival advantages over partial pericardial resection
AVANT GUARD trial extends first-line role for ablation beyond paroxysmal atrial fibrillation
Maintain a high index of clinical suspicion and consider the underlying etiology
Protocol adoption at Cleveland Clinic sharply raised share of transferred patients getting timely PCI