Vaccines, immune checkpoint inhibitors, oncolytic viral therapies and more
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/4ce31d61-9077-47b1-84ff-3bb03f89d832/16-NEU-1934-Ahluwalia-650x450_jpg)
16-NEU-1934-Ahluwalia-650×450
By Manmeet S. Ahluwalia, MD, and Justin D. Lathia, PhD
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
Immunotherapeutic approaches to cancer involve either stimulating the patient’s own immune system to work more efficiently to attack cancer cells or giving the patient synthetic immune system proteins to help mount an immune response to enable killing of cancer cells. Immunotherapy’s potential utility against brain tumors was initially questioned due to the belief that the central nervous system (CNS) was an immune-privileged site, but this belief has since been refuted and there appears to be a dynamic interaction between the peripheral systemic immune system and the CNS.
This is a highly welcome insight, as glioblastoma — the most common primary malignant brain tumor — is still associated with dismal outcomes. Average survival is 15 to 18 months with the traditional treatment paradigms of surgery, chemotherapy and radiation, and less than 10 percent of patients survive more than five years.
Recent years have seen considerable excitement around immunotherapeutic approaches in this patient population, and Cleveland Clinic’s Rose Ella Burkhardt Brain Tumor and Neuro-Oncology Center is participating in a multitude of clinical trials assessing a wide range of these approaches. Those trials are outlined in Table 1 and detailed below.
Unlike some cancers, such as melanoma and kidney cancer, glioblastomas are not inherently immunogenic. One of the challenges has been to induce immune responses to glioblastoma.
One such approach uses a peptide-based vaccine to induce an immune response. Survivin is an intracellular protein that regulates cell division and inhibits apoptosis. In the setting of cancer, high-level survivin expression is associated with poor outcomes and high rates of disease recurrence and therapy resistance. These observations prompted development of SurVaxM, a synthetic long peptide mimic vaccine that stimulates an immune response to survivin.
Advertisement
In the wake of a phase 1 study of SurVaxM demonstrating safety and preliminary efficacy in patients with recurrent malignant glioma, an ongoing phase 2 study is combining SurVaxM with the oral chemotherapy agent temozolomide in patients with newly diagnosed glioblastoma. Cleveland Clinic and Roswell Park Cancer Institute are recipients of a Roswell Park Alliance Foundation grant to support this study.
Additional vaccine-based immunotherapy approaches in trials here and at other centers include:
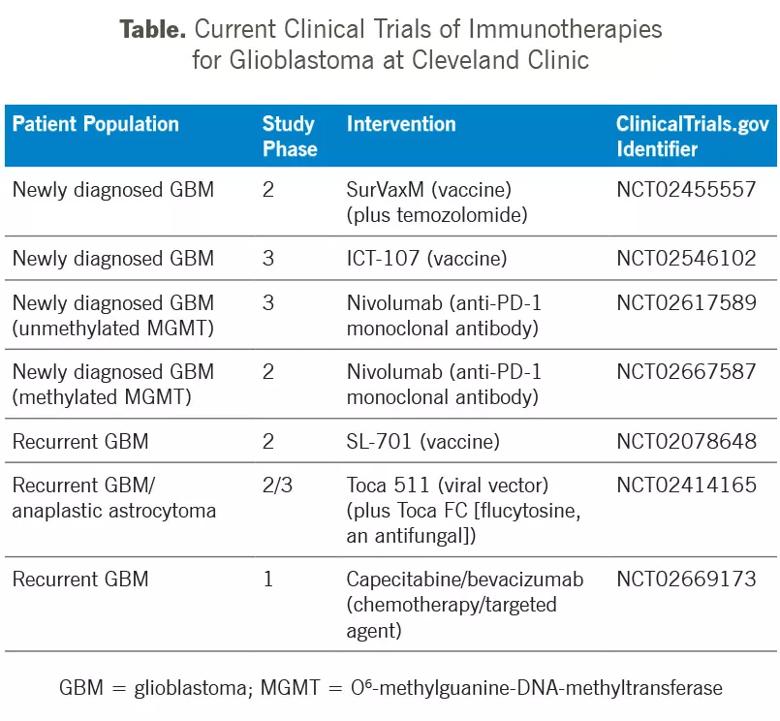
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/6d44ffbb-adf4-4fcc-be4c-20c4ff6240cd/16-NEU-1934-Ahluwalia-Inset_jpg)
Considerable enthusiasm surrounds the use of immune checkpoint inhibitors in glioblastoma. These agents have demonstrated promising activity in melanoma, lung cancer, head and neck cancer, and kidney and other urothelial cancers. They target molecules/pathways that serve as checks and balances on immune responses, and can enable a patient’s T cells to attack cancer cells by releasing the brakes on the immune system.
Two multicenter clinical trials are currently open at Cleveland Clinic investigating the efficacy of one such agent, the anti-PD-1 monoclonal antibody nivolumab, in newly diagnosed glioblastoma patients with either unmethylated O6-methylguanine-DNA-methyltransferase (MGMT) or methylated MGMT.
Advertisement
Oncolytic viral therapeutic-based strategies work on the premise that a modified virus can infect tumors and cause them to self-destruct, thereby facilitating an immune response against the cancer. Viruses being investigated in glioblastoma include poliovirus, genetically engineered poliovirus, the genetically engineered adenovirus known as DNX-2401, measles virus, the retroviral replicating vector Toca 511, and herpes simplex virus.
Of these approaches, the one furthest along in development is Toca 511, which expresses the cytosine deaminase gene and selectively delivers the gene to the tumor. It is being studied in combination with Toca FC, a novel formulation of the antifungal drug flucytosine that gets converted to the anticancer drug 5-fluorouracil within infected cancer cells.
A Cleveland Clinic investigator co-directed a newly published phase 1 study that demonstrated significantly improved survival with these therapies in recurrent high-grade glioma relative to an external control. The study also showed excellent tolerability, and a phase 2/3 randomized, controlled study of Toca 511 and Toca FC in subjects undergoing surgery for recurrent glioblastoma/anaplastic astrocytoma is now accruing patients at numerous centers, including Cleveland Clinic.
Most current immunotherapeutic approaches target immune interactions rather than immunosuppressive cells themselves, so a direct attack on these immunosuppressive cells would represent an attractive treatment strategy.
Myeloid-derived suppressor cells (MDSCs), a heterogeneous class of immature immunosuppressive cells, accumulate in multiple tumor types and suppress cytotoxic immune cells via cytokine secretion. Cleveland Clinic investigators have demonstrated that patients with glioblastoma have elevated MDSCs in blood and tumor, and that these MDSCs produce reversible T-cell dysfunction. In a recent paper, we found that MDSCs localize in tumor regions adjacent to therapeutically resistant tumor cells with stem cell properties (i.e., cancer stem cells). Cancer stem cells produce factors responsible for MDSC function, and targeting these factors can reduce the efficiency of MDSCs.
Advertisement
We also developed a MDSC targeting strategy that relies on low dosing of 5-fluorouracil, a common chemotherapy. In preclinical models, this strategy severely attenuated MDSC numbers and glioblastoma growth while concomitantly increasing cytotoxic T-cell numbers. An ongoing phase 1 trial at Cleveland Clinic is investigating a novel chemotherapeutic strategy targeting MDSC immunosuppression using an alternative dosing schedule of the oral chemotherapeutic capecitabine plus the angiogenesis inhibitor bevacizumab in patients with recurrent glioblastoma. We look forward to sharing results in the coming months and years.
Dr. Ahluwalia is Director of the Brain Metastasis Research Program, Associate Director of Clinical Trials, and Head of Operations in Cleveland Clinic’s Rose Ella Burkhardt Brain Tumor and Neuro-Oncology Center.
Dr. Lathia is Associate Professor in the Department of Cellular and Molecular Medicine in Cleveland Clinic Lerner College of Medicine.
Advertisement
Advertisement
How to use? Consider starting during the acute attack and seek patient preferences for chronic use
Quantitative imaging adds diagnostic value beyond 3T MRI in nearly half of patients
Expert shares insight on intrathecal baclofen pumps to treat spasticity
Opportunities and impacts of a growing surgical approach
Despite safety concerns and mixed trial results, experts see potential for this indication
Experts endorse a push to improve prevention and treatment following repetitive head injury
A population with very high lifetime risk presents care challenges and pathophysiologic insights
Two Cleveland Clinic neurologists review biomarker advances, targeted therapies and unresolved clinical challenges