Cleveland Clinic-led study supports investigation of similar immunomodulatory therapies
Results of the first placebo-controlled study of the monoclonal antibody mavrilimumab in hospitalized patients with COVID-19 pneumonia and heightened systemic inflammation are encouraging enough to merit further investigation. So conclude investigators with the trial, known as MASH-COVID, whose results were published online in Lancet Rheumatology on March 17.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
“The results of this small study did not demonstrate a statistically significant outcomes benefit, but they did show a numeric benefit for patients treated with mavrilimumab and no safety concerns,” says lead investigator Paul Cremer, MD, a Cleveland Clinic cardiologist. “These are encouraging hypothesis-generating findings that are consistent with other studies suggesting a potential role for these therapies in this setting.”
The study, which was funded by Kiniksa Pharmaceuticals, was initiated by Dr. Cremer and coordinated by Cleveland Clinic’s academic research organization, C5Research. It was designed as a small signal-of-efficacy trial to assess whether inhibition of granulocyte–macrophage colony-stimulating factor (GM-CSF) with mavrilimumab would improve clinical outcomes in some of the sickest patients with COVID-19 — those with COVID-19 pneumonia and systemic hyperinflammation. “It was not intended to be definitive for treatment effect,” explains Dr. Cremer.
GM-CSF is a pro-inflammatory cytokine that drives innate immune response and is particularly important in how the lungs respond to inflammatory stimuli. In the first phase of serious COVID-19 cases, characterized by severe viral infection, viral clearance is key, Dr. Cremer notes. “GM-CSF may actually be advantageous in clearing the virus early on,” he says.
The second phase of COVID-19 can be characterized by an overwhelming inflammatory response. “In this phase, GM-CSF becomes a bad actor, perpetuating lung injury,” he continues. “As such, we thought GM-CSF might be a mediator of the hyperactive inflammatory response associated with respiratory failure and death in COVID-19. Mavrilimumab targets GM-CSF, attenuating this overactive inflammatory response.”
Advertisement
Dr. Cremer assembled a consortium of colleagues to discuss the possibility of testing this theory. It was early in the COVID-19 pandemic, and treatments were being promoted with little scientific support. “We wanted to do something to better understand this disease to help improve patient outcomes,” he says. “We felt the best way to help patients would be to study a potential therapy in a randomized controlled trial, as opposed to trying many different things in an uncontrolled way and not knowing what was and was not working.”
Between May 28 and Sept. 15, 2020, 40 patients were enrolled in the trial — 37 of them across Cleveland Clinic hospitals in Northeast Ohio and Florida — and randomly assigned to a single intravenous infusion of mavrilimumab or placebo. Recruitment of 60 patients had been planned, but the trial was stopped early due to slow enrollment after the first surge of COVID-19.
At day 14, 57% of patients in the mavrilimumab group and 47% of patients in the placebo group met the primary endpoint of being alive and off supplemental oxygen therapy (odds ratio = 1.48; 95% CI, 0.43-5.16).
At 28 days, 95% of patients on mavrilimumab and 79% of those on placebo were alive and without respiratory failure. By study completion at day 60, one patient (5%) randomized to mavrilimumab had died, compared with four patients (21%) randomized to placebo.
Adverse events were comparable between the groups, and no safety concerns were seen. “This is very important when giving a treatment to patients who are already critically ill,” says Dr. Cremer. “There was some worry about the effect of so much immunosuppression, but we haven’t seen an increase in secondary infections with this drug.”
Advertisement
Patients who become very ill with COVID-19 pneumonia represent a subset who experience an overactive inflammatory response, Dr. Cremer observes. Because various therapies block immune response through different pathways, mavrilimumab is one of several with potential for treating these very sick patients.
Tocilizumab is an anti-interleukin-6 monoclonal antibody that has some supportive evidence for use in a COVID-19 target population similar to that of mavrilimumab: patients hospitalized with COVID-19 pneumonia with low oxygen levels and high levels of inflammation. “Patients who don’t meet these criteria are not likely to respond,” says Dr. Cremer.
As far as mavrilimumab is concerned, initial results of a larger trial of the agent run by Kiniksa Pharmaceuticals are expected within a couple of months. Additionally, a phase 3 study of lenzilumab, another monoclonal antibody against GM-CSF, just reported top-line results showing significantly greater odds of ventilation-free survival compared with placebo in hospitalized patients with COVID-19, and results from further studies are expected soon.
“I think we’ll know shortly whether targeting GM-CSF with these agents is effective or not,” Dr. Cremer concludes.
Advertisement
Advertisement
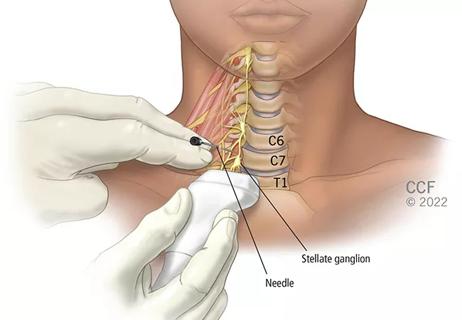
Patients report improved sense of smell and taste
Clinicians who are accustomed to uncertainty can do well by patients

Unique skin changes can occur after infection or vaccine

Cleveland Clinic analysis suggests that obtaining care for the virus might reveal a previously undiagnosed condition

As the pandemic evolves, rheumatologists must continue to be mindful of most vulnerable patients
Early results suggest positive outcomes from COVID-19 PrEP treatment
Could the virus have caused the condition or triggered previously undiagnosed disease?

Five categories of cutaneous abnormalities are associated with COVID-19