Data suggest substantial findings beyond lower field strengths
By Z. Irene Wang, PhD, and Stephen E. Jones, MD, PhD
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
MRI has been demonstrated as a reliable and accurate indicator for many of the pathologic findings underlying epilepsy. It has had a major impact on epilepsy surgery by delineating the extent of the epileptogenic zone. However, approximately 40 percent of the total epilepsy surgery population has a negative or “nonlesional” MRI, even using the 3-tesla (3T) epilepsy protocol. Surgery in the absence of a visible lesion is currently one of the greatest clinical challenges for tertiary epilepsy centers.
In the past few years, it has become increasingly clear that some focal epilepsies initially considered to be MRI-negative, or nonlesional, are not truly nonlesional. In fact, surgical series indicated that in 30 to 50 percent of these patients, histopathologic examination of the resected specimens revealed epileptogenic lesions, mainly focal cortical dysplasia (FCD).1
The low detection rate of FCD by visual analysis stems from the fact that FCD can be quite subtle, with small lesions appearing buried in the complex convexities of the neocortex; some of these lesions have subtle signal changes on a single sequence or very few imaging slices, while others may be obscured due to artifacts. Given the practical constraints of time, MRI readers may miss those FCD lesions, which are only discernable with intense scrutiny. This is especially problematic when noninvasive clinical data, such as seizure semiology and scalp EEG, fail to point to a clear sublobar/lobar area of interest. In the presurgical evaluation of MRI-negative patients, identification of a “missed” FCD lesion may markedly simplify the evaluation process — and likely improve postresective seizure outcomes.
Advertisement
At Cleveland Clinic, ultra-high-field 7-tesla (7T) MRI has been available to patients with epilepsy since 2014 for use as a research study. The principal advantage of 7T MRI over lower-field MRI (3T or 1.5T) is its increased signal strength, which can translate to higher resolution. The latter is the factor most likely to be helpful for patients with subtle epileptic lesions. In our recent study published in NeuroImage, formal assessment of lesion conspicuity among 80 pairs of 7T and 3T images (from patients with a variety of neurological diseases, including epilepsy) found the 7T images to be mildly superior to 3T images overall and clearly superior in many cases.2
Since 2014, 87 patients with epilepsy have undergone 7T MRI at Cleveland Clinic. Research is now underway to evaluate the clinical utility of 7T MRI for enhancing the diagnosis of epilepsy. Of particular interest is the group of 28 patients with negative 3T MRI. Preliminary results showed that visual assessment of these patients’ 7T images revealed additional findings in a total of six patients (21 percent), including subtle FCD in three, FCD with coexisting polymicrogyria in two, and vascular malformation in one.
Our previous research showed that postprocessing of MRI using a morphometric analysis program (MAP) can effectively increase the number of subtle lesions detected.3 We applied postprocessing methods uniquely adapted to 7T MRI on the scans acquired from the 28 patients with negative 3T MRI. Intriguingly, MAP not only reproduced the results of the visual assessment in the majority of cases but also detected subtle FCD abnormalities in an additional 10 patients (36 percent).
Advertisement
The gold standard for determining whether additional abnormalities detected by 7T are true positives or false positives is to examine their correlation with intracranial EEG, histopathology and postoperative seizure outcomes. We investigated all these measures in accordance with the 7T findings, and we continue to follow up with the patients for seizure outcomes.
A total of 11 patients who had additional positive 7T findings (visual or MAP) had already undergone intracranial EEG evaluation. In eight of these patients, the seizure onset zone from intracranial EEG overlapped with the imaging finding. In two patients, the seizure onset zone included the imaging finding but extended to a bigger cortical area. In the remaining patient, the imaging finding was not directly targeted by intracranial EEG, so the relationship could not be assessed.
A total of eight patients who had positive 7T findings (visual or MAP) had already undergone resective surgery. In six of these patients, resection completely included the imaging finding and all patients became seizure-free (five at one-year follow-up; one at six-month follow-up). Histopathology of resected tissue revealed a variety of FCD subtypes, including IIB in two, IIA in one, mild malformation of cortical development with oligodendroglial hyperplasia in two, and malformation of cortical development type II in one. See Figure 1 for an example case. In two patients, resection did not include or only partially included the imaging finding, and both patients had recurring seizures.
Advertisement
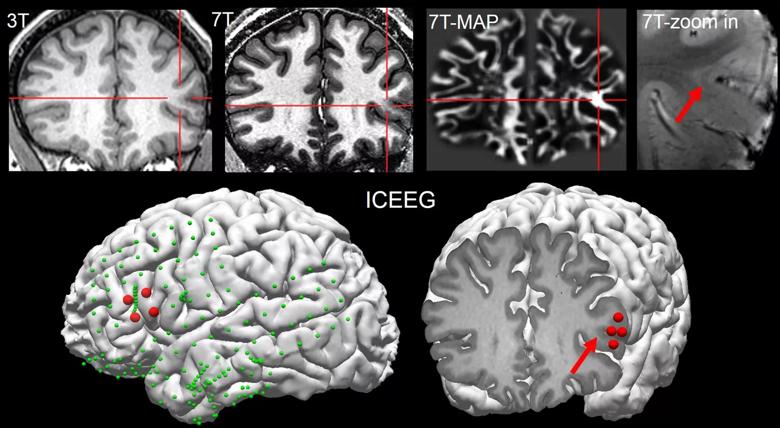
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/6f3f19f1-3810-419a-8bb6-40fbcc1f815b/17-NEU-4236-Wang-inset_jpg)
Figure 1. Imaging studies of a patient in whom both 7T MRI visual analysis and postprocessing analysis (MAP) identified a subtle FCD lesion. 3T MRI was negative. Retrospectively, the lesion (indicated by red crosshair and arrow) can be seen on the 3T MRI, but with much less conspicuity as compared with 7T MRI. Additionally, the lesion was seen by multiple sequences on 7T, including a T1-weighted sequence (top row, second panel) and a T2*-weighted sequence (top row, rightmost panel). The lower row shows the intracranial EEG (ICEEG) findings in this patient, which were highly concordant with the lesion location. All implanted electrodes (subdural grids and depth) are indicated by green spheres; red spheres show those electrodes involved at ictal onset. The lower right panel shows the MRI slice at the exact location of ictal onset (for best viewing effect, only ictal onset is shown on lower right). This patient underwent resective surgery, which completely included the 7T finding; histopathology revealed FCD type IIB; the patient had been seizure-free for six months at most recent follow-up.
Overall, our pilot data suggest that 7T visual and postprocessing analyses have the potential to generate a sizable percentage of additional findings in patients who present with 3T-MRI-negative epilepsy. These additional findings are likely relevant to the epilepsy, given their concordance with intracranial EEG and the pathology support of underlying FCD. Although this is a small, nonconsecutive cohort, these initial results encourage us to pursue further studies of larger number of patients. Longer seizure outcomes also need to be collected.
Advertisement
With our current setup, there is substantial bias field artifact at the inferior basal temporal regions, due to head coil geometry and sinus cavities. This artifact makes it difficult to image lesions located in those areas, and continuing efforts to improve the hardware are needed to alleviate this issue.
Our initial experience showed that 7T MRI is useful for detecting subtle FCD lesions in 3T-negative patients with epilepsy, particularly those with extratemporal lobe epilepsies. Guidance from MRI postprocessing further assists FCD detection. These new noninvasive techniques have the potential to optimize seizure outcomes after epilepsy surgery, and to increase the number of favorable candidates who can be offered potentially curative surgery.
Part of this research was made possible by the JoshProvides Epilepsy Assistance Foundation Research Grant. The authors thank Mark Lowe, PhD, and Sehong Oh, PhD, for their significant technical contributions.
Dr. Wang (wangi2@ccf.org) is a staff scientist in Cleveland Clinic’s Epilepsy Center and joint staff in the Department of Biomedical Engineering, Cleveland Clinic Lerner Research Institute.
Dr. Jones (joness19@ccf.org) is Vice Chair for Research and Academic Affairs in Cleveland Clinic’s Imaging Institute and holds an appointment in the Epilepsy Center.
Advertisement
Despite safety concerns and mixed trial results, experts see potential for this indication
Experts endorse a push to improve prevention and treatment following repetitive head injury
A population with very high lifetime risk presents care challenges and pathophysiologic insights
Two Cleveland Clinic neurologists review biomarker advances, targeted therapies and unresolved clinical challenges
Data-driven segmentation approach shows promise for seizure characterization with utility for clinical decision making
Alzheimer’s studies delve into preventing and modifying the disease’s trajectory and impact
Study finds broadly similar outcomes between MIS and open surgical approaches
System uses clinical data routinely collected at clinical visits