Clinical trial finds supplements no better than standard care in ambulatory patients
Ten days of high-dose zinc, ascorbic acid or a combination of the two has no significant impact on severity or duration of symptoms when taken early in the course of COVID-19.
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
That’s the conclusion of a randomized open-label trial, called COVID A to Z, that compared these interventions with usual care among more than 200 ambulatory patients with COVID-19. The study, which was stopped early for futility, was conducted by Cleveland Clinic researchers and published in JAMA Network Open.
“Based on this study, neither high-dose zinc nor ascorbic acid can be recommended to reduce symptoms of COVID-19 illness,” says the study’s senior author, Milind Desai, MD, Director of Operations in Cleveland Clinic’s Department of Cardiovascular Medicine.
Zinc and ascorbic acid (vitamin C) supplementation have long been thought to have a possible role in fighting viral infection, based on theoretical grounds and some evidence for efficacy. The over-the-counter supplements are taken by many people at the onset of a viral illness in the hope of shortening the disease course. Evidence suggests that zinc increases the ability of polymorphonuclear cells to fight infection, while ascorbic acid’s antioxidant properties play a role in immunity. Some studies have suggested that high doses of these supplements taken early in the course of a common cold may reduce duration and severity of symptoms.
The current study was undertaken to investigate a possible role for zinc and ascorbic acid for ambulatory patients with COVID-19 in reducing symptom severity and duration.
The study enrolled 214 outpatients (mean age, 45.2 ± 14.6 years; 62% female) diagnosed with SARS-CoV-2 infection by PCR assay from Cleveland Clinic outpatient facilities in Ohio and Florida between late April and mid-October 2020. After a positive diagnosis, subjects were randomized to one of the following treatments for 10 days:
Participants tracked their symptoms daily and rated them from 0 to 3, ranging from no symptoms to mild, moderate or severe symptoms. At the beginning of the study, four symptoms were monitored (fever/chills, shortness of breath, cough and fatigue), but the following symptoms were added after July 16 in response to new Centers for Disease Control and Prevention guidance: muscle or body aches, headache, loss of taste, loss of smell, congestion/runny nose, nausea, vomiting and diarrhea.
Patients were contacted weekly until day 28 to collect scores and check on hospitalizations, adverse effects and additional medications. The primary endpoint was days to achieve a 50% reduction in symptoms.
Planned interim analysis at roughly 40% of expected enrollment (214 of 520 patients) revealed the following mean times to the primary endpoint:
The between-group differences were not statistically significant (overall P = 0.45), and the trial’s futility criteria were met for the three active treatment groups versus the usual-care group, prompting study termination.
Moreover, no significant differences were found in any secondary outcomes, which included number of days to reach absence of fever, cough, shortness of breath and fatigue; cumulative symptom severity score at day 5; hospitalizations; and deaths. Fewer than 10% of patients experienced a side effect deemed to be related to supplement therapy, with slightly more seen in the arm receiving ascorbic acid only (nausea, diarrhea and stomach cramps).
According to Dr. Desai, the jury is still out on other potential roles of supplements for COVID-19. Prevention studies are currently being conducted for ascorbic acid, zinc and vitamin D, and whether intravenous ascorbic acid can help critically ill patients is being investigated.
Nevertheless, he notes, the current study has employed a rigorous randomized controlled design to show that over-the-counter (OTC) supplements appear to have no benefit in this outpatient setting that is similar to their widespread use for other viral illnesses.
“The fact that even high-dose ascorbic acid and zinc — more than is typically taken by consumers — did not demonstrate a benefit in this study suggests a clear lack of efficacy for reducing symptom duration and severity in ambulatory COVID-19 patients,” says Dr. Desai. “In the context of the multibillion dollar OTC supplement market, it’s important that patients recognize that taking supplements with no proven benefit can expose them to risk of adverse effects.”
“It is understandable why people want to take supplements to protect themselves against COVID-19,” adds Leslie Cho, MD, Co-Section Head of Preventive Cardiology. “However, there are no data to date showing that these supplements help. In contrast, sticking to a balanced diet, exercising and a getting a good night’s sleep are actions that can help.”
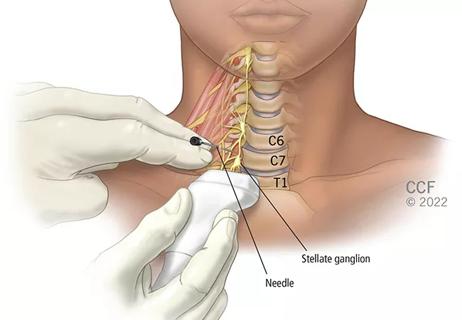
Patients report improved sense of smell and taste
Clinicians who are accustomed to uncertainty can do well by patients
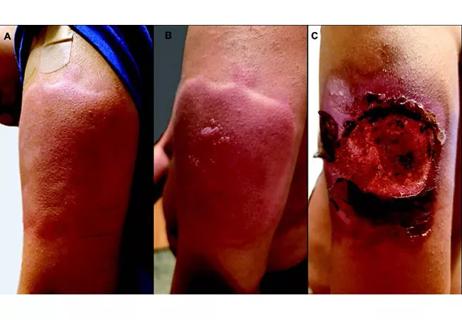
Unique skin changes can occur after infection or vaccine

Cleveland Clinic analysis suggests that obtaining care for the virus might reveal a previously undiagnosed condition
As the pandemic evolves, rheumatologists must continue to be mindful of most vulnerable patients
Early results suggest positive outcomes from COVID-19 PrEP treatment
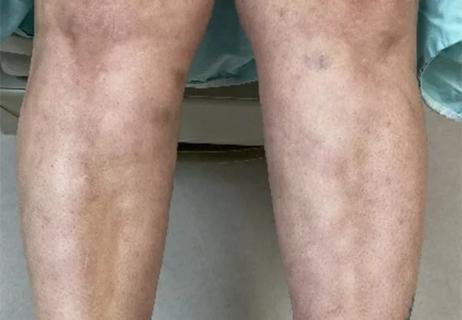
Could the virus have caused the condition or triggered previously undiagnosed disease?
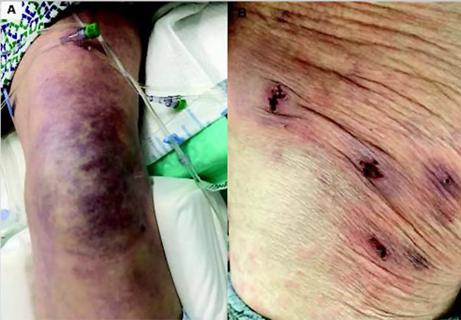
Five categories of cutaneous abnormalities are associated with COVID-19