This approach led to local control for patients with brain metastases more than 2 cm in diameter
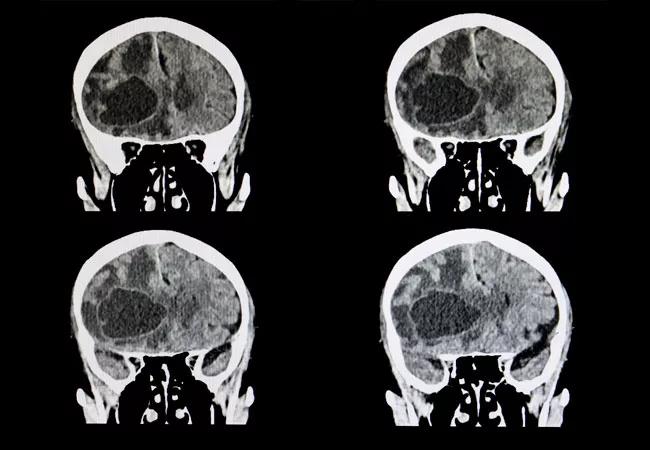
A research effort, led by Cleveland Clinic experts, demonstrated that preoperative stereotactic radiosurgery (SRS) with dose escalation followed by surgical resection for brain metastases larger than 2 cm led to local control that is similar to standard approaches (i.e., postoperative SRS or whole-brain radiation therapy), and with potentially fewer long-term side effects.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
These findings, “Results of a Phase 1 Trial of Dose Escalation for Preoperative Stereotactic Radiosurgery for Patients with Large Brain Metastases” were recently presented by Erin Murphy, MD, Associate Professor, Department of Radiation Oncology, Cleveland Clinic, during the ASTRO 2023 annual meeting.
“Single session stereotactic radiosurgery alone for certain brain metastases (>2 cm in diameter) typically leads to suboptimal local control,” says Dr. Murphy. “Additionally, surgical resection alone also results in unreliable local control and perioperative radiation is needed. We initiated this prospective, phase 1 trial to determine the safety of preoperative SRS at escalating doses followed by surgical resection.”
This study included three radiosurgery dose cohorts based on maximum tumor diameter of the index lesion: 18 Gy for >2-3 cm, 15 Gy for >3-4 cm and 12 Gy >4-6 cm. Concurrent stereotactic radiosurgery alone to other smaller lesions was permitted using standard RTOG dosing.
“Within each of these cohorts, we started at the base dose that we use for intact tumor dosing and escalated the dose at three gray (Gy) increments,” says Dr. Murphy. “If at least two patients enrolled safely at the base dose, we would escalate. If there was one dose limiting toxicity, we would continue to enroll until the time of escalation unless there was a second dose limiting toxicity. In those cases, the previous, lower dose was determined to be the maximum safe dose.”
The researchers defined dose limiting toxicity as grade 3 or greater acute toxicity within three to four months following SRS. Within two weeks of SRS, enrolled patients underwent surgical resection. They were then followed by the study authors with image and neurological evaluations every three months.
Advertisement
This analysis included 35 patients with a median age of 63 years. The median interval between SRS and surgery was two days. Among enrolled patients, the most common histology was non-small cell lung cancer (57.1%), followed by breast cancer (14.3%).
Patients with a tumor size of >2-3 cm were enrolled up to the second dose level (21 Gy). The third dose level was reached for patients in the >3-4 cm and >4-6 cm cohorts (21 Gy and 18 Gy, respectively).
Study authors observed a total of three dose limiting toxicities. Two occurred in the >3-4 cm cohort (grade 3 wound dehiscence and grade 3 wound infection) and one in the >4-6 cm group (grade 4 hematoma).
“We determined that the maximum tolerable dose was 18 Gy (2nd dose level) for the >3-4 cm cohort, and 18 Gy (3rd dose level) for the >4-6 cm group,” says Dr. Murphy, while discussing their key findings.
“At two years, the rate of leptomeningeal disease was 0%,” she reports. “This suggests a significant benefit over post-operative radiosurgery, which demonstrates a higher rate of leptomeningeal disease. Additionally, we observed good local control after long-term follow up. At a median median follow-up of 64 months, the six- and 12-month local control rates were 88.8% and 79.1%, respectively.”
Dr. Murphy and her colleagues also found that preoperative stereotactic radiosurgery with dose escalation followed by surgical resection led to six- and 12-month distant brain control of 63.1% and 55.3%, respectively. Overall survival at six and 12 months was 82.9% and 59.0%.
Advertisement
Based on these phase 1 findings, further investigation of this approach is warranted, according to Dr. Murphy. The research team has opened the phase 2 arm of this trial, and enrollment is ongoing. The study will be conducted at the maximum tolerated doses, and patients will be followed for efficacy and toxicity outcomes.
“Preoperative SRS with dose escalation followed by surgical resection has the potential to benefit a variety of patients with brain metastases,” says Dr. Murphy. “In this era of more molecular-targeted therapies and immunotherapies, patients who have brain metastases are living longer. Finding approaches that give patients, especially those who present with large tumors, durable control is important.”
Advertisement
Advertisement

Largest study to date comparing direct-acting oral anticoagulants to low-molecular-weight heparin

Research demonstrates improved overall survival for patients receiving comprehensive treatment for breast cancer in addition to radiation or surgical intervention for brain cancer
Improvements enable targeting of brain tumors with single-session, fractionated or neoadjuvant approaches
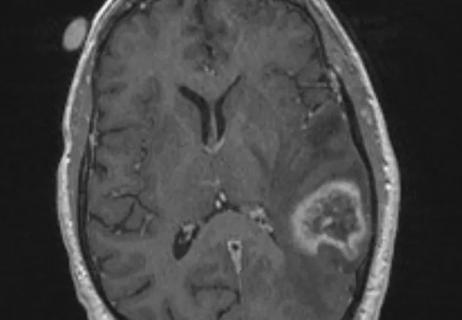
We now have an expanded toolbox to extend survival and lessen treatment toxicity

Large retrospective study suggests DOACs are safe, effective alternative to low-molecular-weight heparin in complex patient population

Advances in genomics, spinal fluid analysis, wearable-based patient monitoring and more

An update on the technology from the busiest Gamma Knife center in the Americas

Radiation oncology department finds weekly plan of care meetings have multiple benefits