Ongoing work is looking for exact mechanism
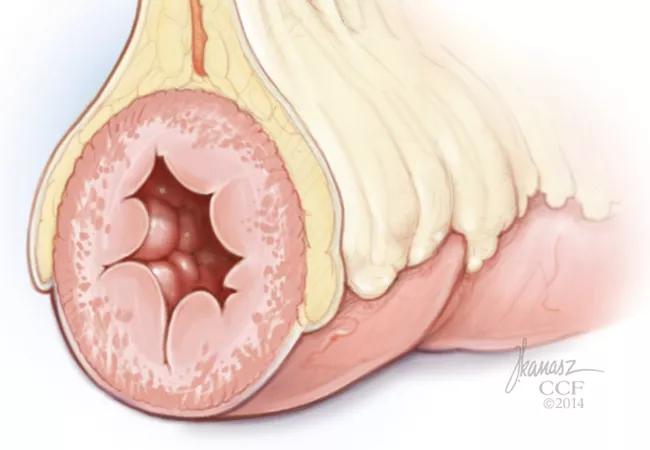
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/37495b1d-09fe-4910-b9fd-1e4ed1d2c64b/18-DDI-397-Rieder-Hero-Image-650x450pxl_jpg)
18-DDI-397-Rieder-Hero-Image-650x450pxl
More than 80 percent of patients with Crohn’s disease who develop intestinal strictures have creeping fat — mesenteric fat that wraps around the intestine — in the same location.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
This has led researchers to wonder if there might be a link between the two that, if better understood, might lead to therapeutic interventions. Currently there is no anti-fibrotic therapy available for Crohn’s patients with strictures; most have to undergo interventions with endoscopy or surgery to treat the problem.
“We feel there’s a connection between the two,” says Florian Rieder, MD, of the Department of Gastroenterology and Hepatology. “The creeping fat is in direct contact with the intestinal muscle and when the intestinal muscle thickens then the lumen narrows and patients obstruct.”
Dr. Rieder conducted a study looking at creeping fat derived free fatty acids (FFA) and their role in the development of strictures in patients with Crohn’s disease. He presented the findings Monday at DDW 2018.
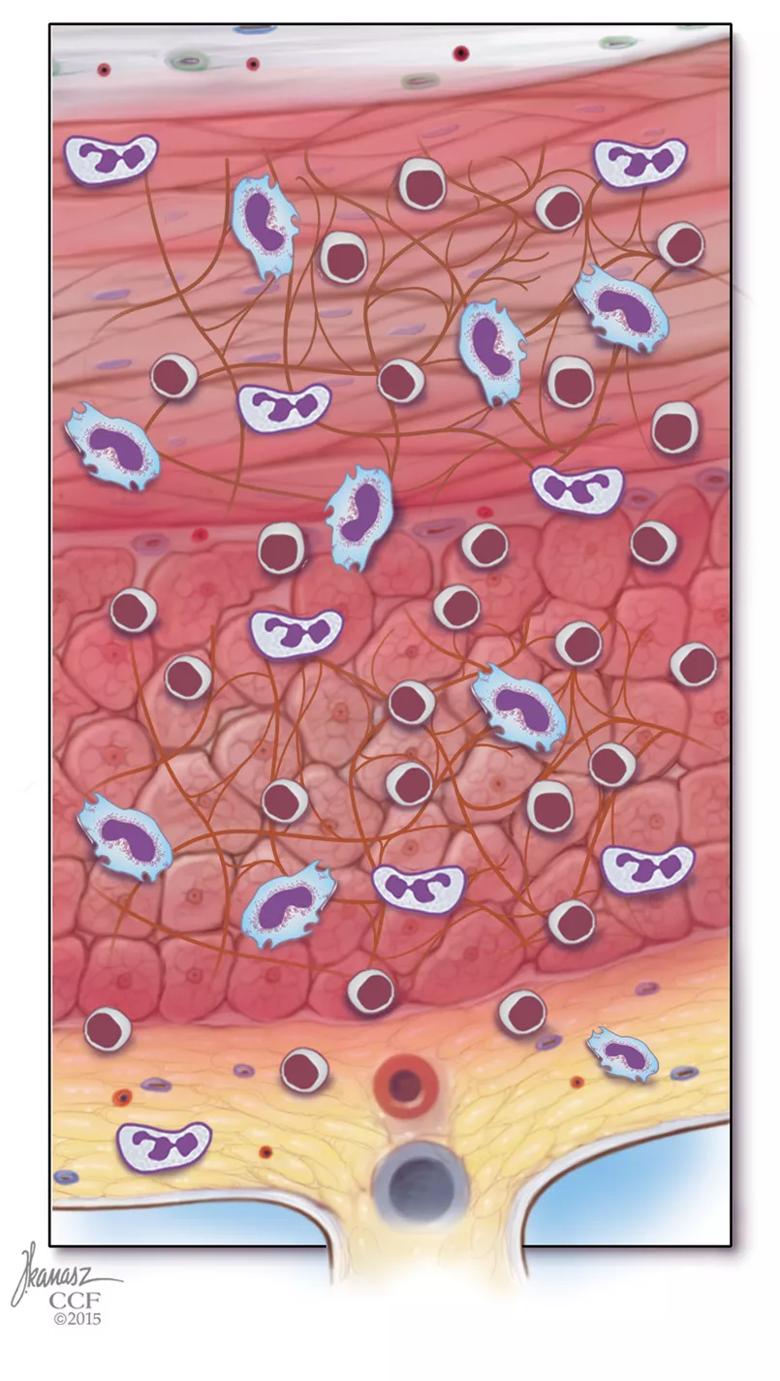
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/2b28c88e-c6f1-46bf-9b7e-2d9f76f7d5e8/18-DDI-397-Chrones-Inset-805pxl-width_jpg)
After culturing creeping fat cells, Dr. Rieder and his colleagues used mass spectrometry to determine which FFA are secreted from the fat cells. They found the FFAs came in a variety of different lengths: palmitate, stearate, oleate, decanoate, butyrate. From there, they exposed intestinal smooth muscle cells from patients with Crohn’s disease to those FFAs, as well as to different adipokines and to TNF, IL-1β, TGF-β1 or bFGF.
“We looked at what fatty acids are secreted by the creeping fat,” Dr. Rieder says, “then we took these fatty acids and we put them on the muscle to see if the muscle proliferates and it did.”
The researchers compared intestinal specimens of patients with Crohn’s disease who had creeping fat and those that did not, as well as patients with ulcerative colitis and healthy controls. They found that creeping fat secreted higher amounts of total, saturated and poly-unsaturated FFA with higher levels of the long-chain FFA palmitate, oleate and stearate (P < 0.05).
Advertisement
In addition, they found that long-chain (but not medium- or short-chain) FFA increased proliferation in smooth muscle tissue and fibroblasts, but not in intestinal epithelial or endothelial cells, and not in preadipocytes or adipocytes. They also found the proliferation was dependent on the signaling pathways p38MAPK, PKC and PI3K, but not MyD88, TLR2, TLR4, NFΚB or CD36.
“So there was significant specificity,” Dr. Rieder says. “Creeping fat derived long-chain but not shorter-chain fatty acids led to proliferation of muscle cells and fibroblasts but not other cells of the intestine.”
The investigators also inhibited FFA uptake into the smooth muscle cell mitochondria by blocking carnitine palmitoyltransferase, CPT-1. This reduced palmitate-induced proliferation in five out of eight smooth muscle cell lines taken from patents with Crohn’s disease. It did not reduce proliferation in the patients with ulcerative colitis or in the healthy controls. By comparison, blocking sphingosine biosynthesis inhibited palmitate-induced proliferation in all of the smooth muscle cells lines.
Finally, the researchers found exposure of the smooth muscle cells to whole mesenteric fat dramatically upregulated smooth muscle cell proliferation and fat-conditioned medium derived from cells of patients with Crohn’s disease significantly increased proliferation twofold compared with patients with ulcerative colitis or to the healthy controls.
“Blocking CPT-1 is one way we may be able to intervene medically,” Dr. Rieder says. “The next step is to find the exact mechanism of how this is mediated.”
Advertisement
To do that, the investigators will label the FFA and use mass spectrometry to determine where they end up inside the smooth muscle cells. “The FFAs can go into many different compartments within a cell and we want to know exactly which ones,” he says. “That will allow us to better determine exactly what they do.”
Advertisement
Advertisement
First full characterization of kidney microbiome unlocks potential to prevent kidney stones
Researchers identify potential path to retaining chemo sensitivity
Large-scale joint study links elevated TMAO blood levels and chronic kidney disease risk over time
Investigators are developing a deep learning model to predict health outcomes in ICUs.
Preclinical work promises large-scale data with minimal bias to inform development of clinical tests
Cleveland Clinic researchers pursue answers on basic science and clinical fronts
Study suggests sex-specific pathways show potential for sex-specific therapeutic approaches
Cleveland Clinic launches Quantum Innovation Catalyzer Program to help start-up companies access advanced research technology