Locations:
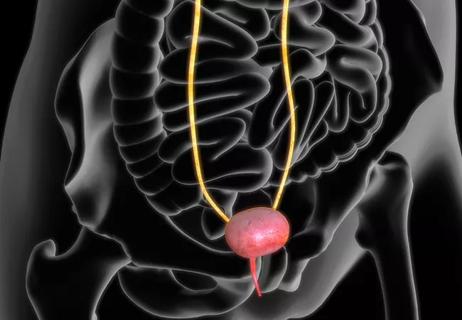
Bimagrumab increases pelvic muscle volume in rat models
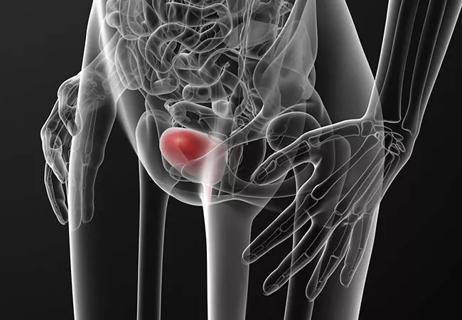
Cleveland Clinic joins national trial to investigate treatment for urinary incontinence

Recall does not necessarily mean transvaginal mesh is defective
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
Advertisement