Paucity of models hinders development of treatments for this lethal disease
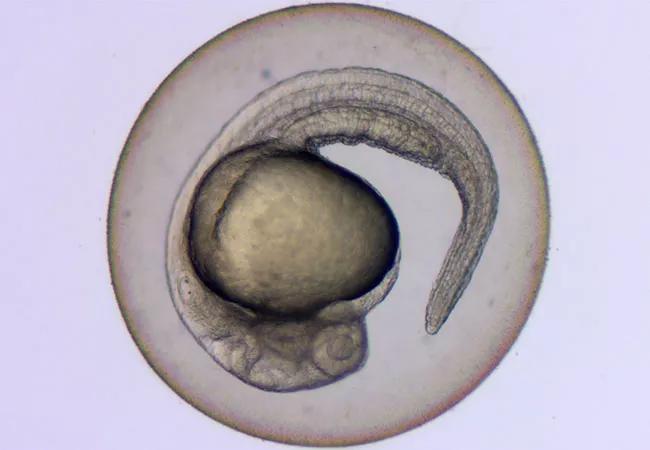
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/b2691ec3-f72e-472d-b864-d2215d32a122/650x450-Ewing-sarcoma_jpg)
Zebrafish embryo
Ewing sarcoma (EWS) is a common and deadly pediatric cancer involving bone and soft tissues with a five-year survival rate of less than 30% for patients with metastatic disease. Patients typically present as adolescents or young adults with advanced disease, according to pediatric oncologist Seth J. Corey, MD, MPH, Professor of Pediatric Hematology, Oncology and Bone and Marrow Transplantation at Cleveland Clinic Children’s, and Molecular Medicine Cancer Biology at Cleveland Clinic.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
The vast majority of patients with EWS have a chromosomal translocation that encodes a EWSR1/FLI1 fusion protein, which spurs the development of tumors. In addition, this disease is notable for recurrent mutations in the TP53 gene, a vital mediator of DNA damage repair, and the STAG2 gene, which is an important component of the cohesion complex associated with DNA replication.
“Unlike other pediatric cancers, there are only a few good models to study EWS and few effective therapies,” he says. “A five-drug cytotoxic regimen has been the mainstay of treatment for the past 30 years.”
With a grant from VeloSano Kids, however, Usua Oyarbide, PhD, of the departments of Pediatrics and Cancer Biology in the Cleveland Clinic Lerner Research Institute, is generating a novel zebrafish model to be used in studying the disease. “We received a one-year grant to establish a transgenic EWSR1/FLI1 zebrafish model that co-expresses mutations in TP53 and STAG2, and then validate that model by testing it against standard drugs like doxorubicin and ifosfamide at different concentrations to see how EWS cell proliferation, migration and response are impacted,” she explains. The study is slated to begin in August or September of this year. Subsequently, the team will request more funding, and hopes to study experimental chemotherapy agents recommended by Peter Anderson, MD, PhD, and Rabi Hanna, MD, staff physicians in the Department of Pediatric Hematology, Oncology and Blood and Marrow Transplantation who are also providing patient samples for the study.
Advertisement
Zebrafish are an excellent model for the study of EWS, sharing a similar genetic structure to humans, as well as many of the same organs and tissues. In addition, Dr. Oyarbide has experience with the zebrafish model, having successfully created other zebrafish models for inherited bone marrow failure syndromes, such as Shwachman-Diamond syndrome. “Cleveland Clinic’s zebrafish aquatic facility consists of two rooms and one quarantine room at the Lerner Research Institute,” she reports. “In my dedicated room, I have more than 400 tanks, and I already possess and am breeding mutant and transgenic lines.”
Zebrafish are tropical fish that are native to southeast Asia, and grow to approximately 1 inch in length. They are almost transparent, so their organs can be visualized, and when human cells expressing EWS are injected into the fish, they can be readily seen under a low-power microscope. Zebrafish typically lay up to 100 eggs at a time at weekly intervals, which mature quickly in five days’ time. They are also much cheaper to maintain than murine models, which are the usual animal models employed. “Other models have disadvantages in that they take a long time to breed and develop cancer, and only produce a few offspring at a time, which means we don’t have the numbers needed to power a study,” Dr. Corey says. Whereas it might take three years to get results with a murine model, the experimental period with zebrafish can be reduced significantly. “Use of zebrafish may reduce the drug development period that can take as long as 10 years to just three years,” he reports.
Advertisement
“Fish can also be genetically manipulated more easily and quickly than murine models,” adds Dr. Oyarbide, “and drugs can be more easily administered.” In addition, there is no need for immunosuppression with fish, since zebrafish do not develop an adaptive immune response during the first four weeks of life.
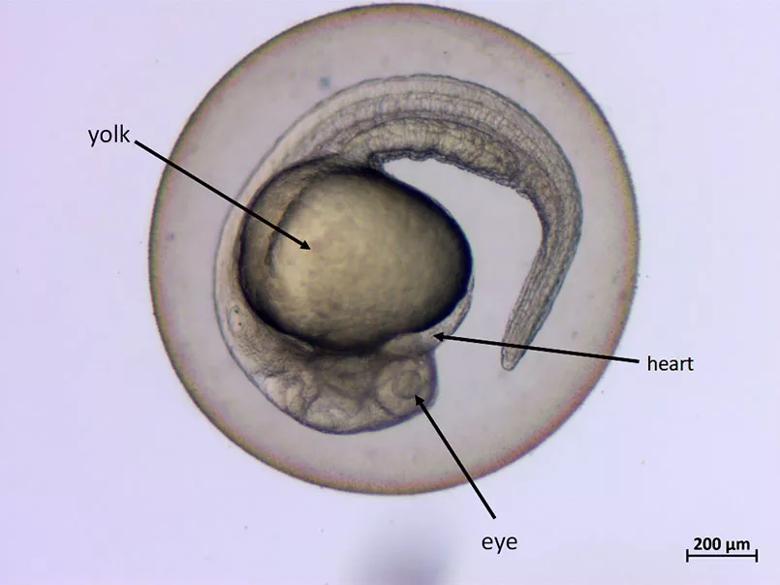
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/bf9836bb-7a9d-4ebf-8e47-221fba85be99/805x-Inset-Ewing-sarcoma_jpg)
Zebrafish model, 1 day-old embryo
Dr. Oyarbide has developed two aims for the study, with the first being to create genetically engineered strains of zebrafish that express EWSRS1/FLI1 in the pursuit of an optimal zebrafish genetic model for ES. She expects these fish to develop EWS tumors by four months of age, at which point she will examine their histopathology. The second aim is to optimize EWS xenografting in zebrafish and assess the efficacy of known antineoplastic agents. “I will perform patient-derived xenografts to study tumor invasion and metastatic dissemination of three different human ES lines (Hs822T, Hs863T and RD-ES),” she says.
“The VeloSano grant will allow us to study EWS biology and provide a strong basis for future experiments that will permit us to study real-time patient tumor aggressiveness and drug responses in models, which is better than in cell lines,” Dr. Oyarbide says. In addition, she hopes the grant will allow her to present her findings at the American Association of Cancer Research annual meeting in 2021.
Please note: This research is funded by a grant from VeloSano Kids. VeloSano Kids was launched in the spring of 2018 as a way for children to get involved with the mission of VeloSano. VeloSano Kids is a year-round, community-driven fundraising initiative, where 100% of the dollars raised supports lifesaving pediatric cancer research at Cleveland Clinic Children’s.
Advertisement
Advertisement
First full characterization of kidney microbiome unlocks potential to prevent kidney stones
Researchers identify potential path to retaining chemo sensitivity
Large-scale joint study links elevated TMAO blood levels and chronic kidney disease risk over time
Investigators are developing a deep learning model to predict health outcomes in ICUs.
Preclinical work promises large-scale data with minimal bias to inform development of clinical tests
Cleveland Clinic researchers pursue answers on basic science and clinical fronts
Study suggests sex-specific pathways show potential for sex-specific therapeutic approaches
Cleveland Clinic launches Quantum Innovation Catalyzer Program to help start-up companies access advanced research technology