For patients with moderate to severe OSA who are intolerant of traditional CPAP therapy, HNS may provide a welcome improvement to QoL
written by Sunjeet Kaur, MD and Reena Mehra, MD, MS
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
Obstructive sleep apnea (OSA), characterized by partial or complete airway obstruction during sleep reflecting apneas and hypopneas respectively, afflicts nearly 1 billion individuals worldwide.1 The standard treatment for OSA is continuous positive airway pressure (CPAP) therapy which serves as a pneumatic splint to achieve airway patency. Although CPAP adherence is improving (range: 50-70%), adherence remains a challenge due to a variety of reasons including, mask interface issues, pressure intolerance, claustrophobia and nasal congestion.2 Hypoglossal nerve stimulation (HNS) is an innovative therapeutic surgical approach that is increasingly used for patients with severe OSA who are intolerant of PAP therapy.
The Cleveland Clinic Sleep Disorders Center has been identified as a Center of Excellence for placement of the current commercially available HNS system (Inspire Medical Systems) leveraging cross-disciplinary collaborations involving Otolaryngology, Pulmonary and Neurology. The Center has conducted 194 implantations (2015-2022).
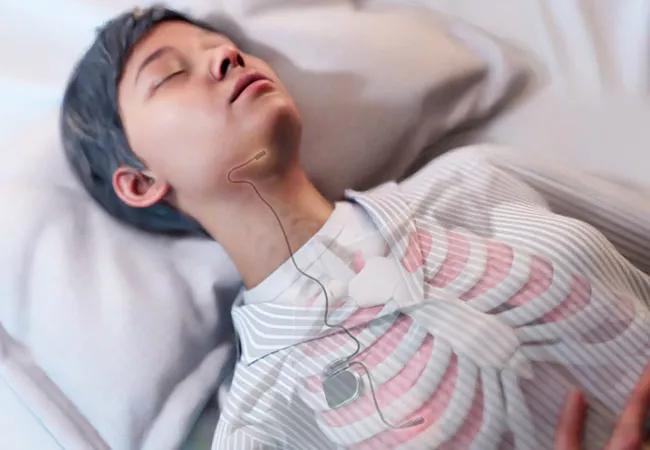
The surgery for implantation is performed as an outpatient procedure and is done under general anesthesia. The device is comprised of three implanted elements: a respiratory sensor, a small implanted electrical impulse generator and a hypoglossal nerve stimulation lead (Figure 1). The system is activated by a hand-held remote control device for nightly use. Unilateral electrical stimulation of the hypoglossal nerve synchronized to respiration produces tongue protrusion. The genioglossus muscle innervated by the hypoglossal nerve is the primary tongue protrusor muscle and dilator of the upper airway. Electrical stimulation of the hypoglossal nerve leads to an increase in the genioglossal muscle tone and a slight protrusion of the tongue. The forward tongue movement and its increased muscle tone along with coupled movement of the soft palate results in an opening of the upper airway, thereby preventing its collapse through stabilization.
Advertisement
Effectiveness and adherence with the device are well-documented. However, selecting the most suitable patients and therapeutic tongue stimulation parameters is crucial for producing the best therapeutic outcomes. Criteria for candidacy include unsuccessful use of CPAP, apnea-hypopnea index (AHI) between 15 and 65, at least 22 years of age and a body mass index (BMI) < 35 kg/m2. In the international ADHERE registry, we identified that increased age, reduced BMI and female sex were predictors of HNS response defined by a reduction in AHI.3 Anatomical evaluation via drug-induced sleep endoscopy (DISE) examination is also an essential step to assess candidacy, i.e., to establish the absence of complete velopharyngeal concentric collapse.
Clinical use of HNS was evaluated in a multicenter, prospective cohort with 126 participants with a mean age of 54.5 years, 83% male, mean BMI of 28.4 kg/m2 and mean baseline AHI of 34. HNS resulted in significant improvements in median AHI at 12 months. This resulted in a 68% reduction, from 29.3 to 9.0 events per hour (p < 0.001).5 In the randomization phase, 46 consecutive participants with an adequate response to therapy were enrolled. The 23 participants randomized to the therapy-maintenance group at 1 week did not have a significant change in AHI compared to a significantly higher AHI observed in the therapy-withdrawal group (25.8 versus 7.6 respectively at 1-week follow-up, p < 0.001). Epworth Sleepiness Scale and Functional Outcomes of Sleep Questionnaire scores were also improved. The only complication specific to the mechanism of the device was transient tongue weakness which occurred in 17% of study participants. HNS therapy adherence, defined as daily use, remained high (81%).
Advertisement
Our group has shown that HNS also improves Insomnia Severity Index at 1 year with results comparable to PAP therapy.4 We have also found that compared to PAP therapy, HNS had a lesser impact on blood pressure improvement and greater improvement of sleepiness symptoms.5 The percentage of non-white patients receiving HNS implants is low underscoring the need to identify barriers and expand access to the non-white patient population.6
HNS is approved for moderate to severe OSA in PAP-intolerant patients without concentric collapse of the soft palate and this option should be included in routine discussions of alternate therapies for OSA in cases of unsuccessful CPAP use. Of the available outcome data, HNS therapy is effective not only to reduce the severity of OSA, but it also improves patient-reported outcomes of daytime sleepiness and quality of life.
Advertisement
Advertisement

A deep dive into the evolution of surgical sleep therapy

Cleveland Clinic study finds that durable weight loss is key to health benefits

Novel procedures provide options for patients who can’t tolerate CPAP

A look at how custom-fitted oral appliances work and when they’re a good fit for patients

Screening tool helps flag conditions that warrant further attention

Patients with epilepsy should be screened for sleep issues

Study shows comparable success rates to tongue base dependent epiglottic collapse

Looking at short-term outcomes in a high-risk population