Significant reduction in risk for vision loss and disease progression
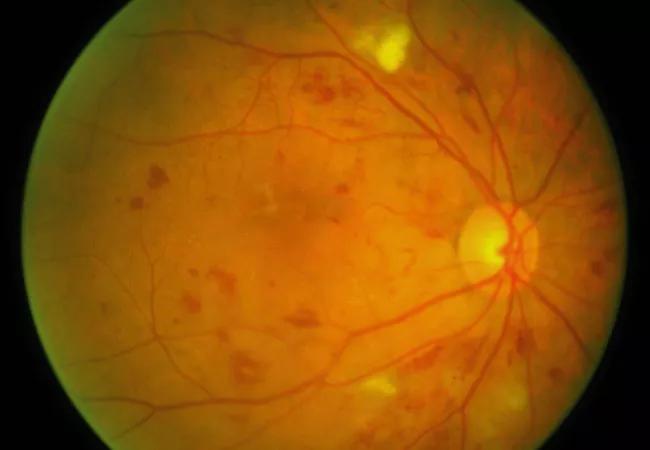
Intravitreal injections of the anti-vascular endothelial growth factor (VEGF) drug aflibercept induce regression of disease in patients with moderately severe to severe nonproliferative diabetic retinopathy (NPDR) and without diabetic macular edema (DME), according to new results of the PANORAMA trial.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
Rishi Singh, MD, of Cleveland Clinic’s Cole Eye Institute, presented the results of the phase 3 randomized, double-blind trial, the first large, prospective trial of its kind since the Early Treatment Diabetic Retinopathy Study, at the American Academy of Ophthalmology 2019 annual meeting.
VEGF, a signal protein, stimulates blood vessel formation. Its overexpression is believed to drive the process of vascular proliferation in diabetic retinopathy. The previously published VISTA and VIVID studies showed that VEGF inhibitors decrease the rate of disease progression in patients with NPDR and DME, but anti-VEGF efficacy and safety had not been studied in patients with NPDR without concurrent DME.
“The last time we studied these patients, laser treatment was the only option,” says Dr. Singh, a retina specialist and Director of Cole Eye Institute’s Center for Ophthalmic Bioinformatics. “Now we have evidence that a treatment regimen achievable in the real world works in these high-risk patients.”
The PANORAMA study randomized 135 participants with moderately severe to severe NPDR (Diabetic Retinopathy Severity Scale [DRSS] scores from 47 to 53) and without DME to receive intravitreal aflibercept injections every 16 weeks after four loading doses, 134 participants to receive the same injections every eight weeks after five loading doses, and 133 participants to a sham control. Noncontrols had type 1 or 2 diabetes mellitus and a baseline best-corrected visual acuity (BCVA) score of ≥ 69 letters. The mean age was 55.7 years, and 44% of patients were women.
Advertisement
The primary endpoint was the proportion of patients whose DRSS scores improved two or more steps in one year. Researchers also sought to determine whether aflibercept reduced vision-threatening events and impacted visual acuity.
At week 52, 65.2% of the 16-week injection group and 79.9% of the eight-week group had a 2-step or more improvement in their DRSS scores versus 15% of the sham group (P < 0.0001 for both). During the study, 20% of control patients developed a vision-threatening complication or DME, compared with only 4% of the 16-week treatment group and 3% of the eight-week group (P < 0.0001 for both).
“These are excellent outcomes for patients who are at risk for vision loss,” says Dr. Singh. “An 80 to 90% reduction in risk for threats to vision is great news for patients and may result in a new treatment paradigm for patients. However, these treatments come at a cost and potential risk and DME can be very effectively managed if it develops. More information on the long-term importance of improving DRSS scores are needed before this paradigm is likely to be widely adopted in clinical practice.” The DRCR Retina Network is conducting a 4-year study (Protocol W) that should help to answer this question.
Diabetic retinal disease is the leading cause of blindness in working-age adults in the United States. Dr. Singh’s work to establish effective treatments for DME and diabetic retinopathy earned him the 2018 early career investigator award from the President of the American Society of Retina Specialists. He is President of the Retina World Congress and leads the Cole Eye Institute Center for Ophthalmic Bioinformatics, which addresses leading causes of blindness.
Advertisement
“Understanding presentations of these ophthalmic diseases, their risk factors, their prognosis for progression and their optimal response to treatment through bioinformatics processes is an important step at improving the lives of our patients,” says Dr. Singh.
“PANORAMA results are very encouraging,” he notes. “But there is always more work to do. We hope that harnessing the power of bioinformatics will allow further insight into many ophthalmic diseases, including diabetic retinal disease.”
Photo: ICEH.
Advertisement
Advertisement

Motion-tracking Brillouin microscopy pinpoints corneal weakness in the anterior stroma

Registry data highlight visual gains in patients with legal blindness

Prescribing eye drops is complicated by unknown risk of fetotoxicity and lack of clinical evidence

A look at emerging technology shaping retina surgery

A primer on MIGS methods and devices

7 keys to success for comprehensive ophthalmologists

Study is first to show reduction in autoimmune disease with the common diabetes and obesity drugs

Treatment options range from tetracycline injections to fat repositioning and cheek lift