Novel use of tPA reduces fibrin response in the eye
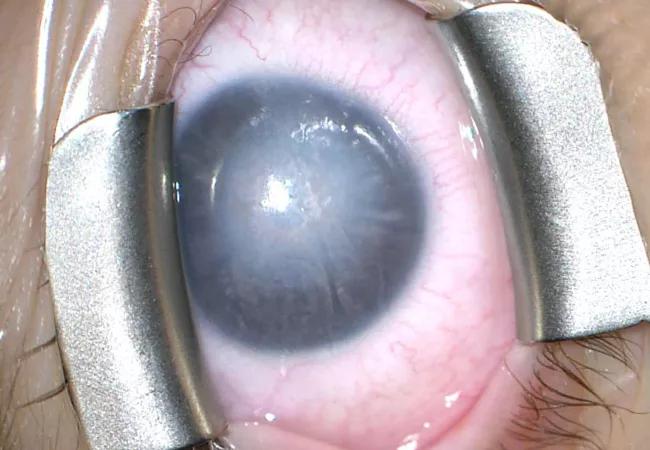
Above: A young patient presents with significant diffuse corneal haze later diagnosed as posterior polymorphous corneal dystrophy.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
A 16-month-old patient with watery eyes and light sensitivity presented to cornea and external disease specialist Jeffrey Goshe, MD, at Cleveland Clinic Cole Eye Institute. The patient had been diagnosed with congenital nasolacrimal duct obstruction when 6 months old. Corneal dystrophy had been suspected as well, but it was not severe enough to warrant further treatment at the time.
Over the previous year, the patient’s parents had begun noticing the child’s increasing sensitivity to light and squinting, both of which were partially alleviated with sunglasses. In addition, the child held objects, including a mobile tablet and other screens, close to his face.
During an ocular exam under anesthesia, the patient was found to have significant diffuse corneal haze in both eyes. The corneal thickness in both eyes was more than twice that of normal corneas (>1,100 microns vs. 550 microns), indicating the presence of severe corneal edema. Due to these clinical findings and a family history of posterior polymorphous corneal dystrophy (PPCD), the child was diagnosed with PPCD.
PPCD is a rare genetic disease affecting the corneal endothelium and Descemet’s membrane. The disorder affects approximately 0.1% or less of the U.S. population.
“Most patients with PPCD are diagnosed in adolescence or adulthood,” says Dr. Goshe. “Few are diagnosed in early childhood. This was an uncommon presentation of an uncommon disease.”
Many cases of PPCD are asymptomatic, with only patches of irregular cells, described as “snail tracks,” incidentally identified on slit lamp examination. PPCD typically progresses slowly over years or decades, and many patients never require treatment.
Advertisement
Symptomatic cases with mild corneal edema can be managed with a topical osmotic, such as hypertonic saline, to desiccate fluid from the cornea. However, for more severe cases, transplanting the corneal endothelium is the only definitive treatment.
“This patient needed surgery urgently due to the severity of his presentation and his young age,” says Dr. Goshe. “Impaired vision during early childhood causes amblyopia, a permanent reduction in long-term visual potential. The younger the age, the faster it happens. When vision is poor, the developing brain will rapidly start losing the ability to create the necessary neurological pathways to achieve normal vision.”
The endothelium is a thin, clear layer on the inside of the cornea.
“If you think of the cornea as the window into the eye, you would have to be ‘inside the house and touching the window’ to touch the layer of defective cells in patients with PPCD,” explains Dr. Goshe.
Surgery for PPCD involves removing this thin layer of cells and replacing it with tissue procured from the healthy cornea of a deceased donor. The transplanted tissue is a circular piece, about 0.3 inches in diameter and thinner than a strand of hair. It is so delicate that it can be handled only with the smallest surgical instruments, and only minimally to avoid damaging the cells.
“The endothelium in a healthy eye prevents excess fluid from accumulating in the cornea,” says Dr. Goshe. “Proper hydration creates the perfect spacing of collagen fibers, which makes the cornea transparent. When the cornea has too much fluid, swelling disrupts this spacing, leading to haze and, therefore, blurry vision.”
Advertisement
A very swollen cornea, in which fluid percolates to the surface, can result in bullae. This is what causes pain and light sensitivity, as with the patient in this case.
Historically, the only surgery to replace corneal endothelium was akin to removing and replacing an entire window just to peel off and reapply plastic wrap on the opposite side, explains Dr. Goshe. This surgery, penetrating keratoplasty, typically required 16-24 sutures to join the new tissue to the patient’s cornea. The introduction of lamellar (“partial thickness”) corneal transplants, such as Descemet’s stripping automated endothelial keratoplasty (DSAEK) and Descemet’s membrane endothelial keratoplasty (DMEK), changed that.
“Today we don’t remove the entire cornea, we just peel off the old endothelium and roll out a new one,” he says.
DMEK in pediatric patients is challenging because children’s eyes are more elastic, says Dr. Goshe. It’s harder to maintain spacing and complete tasks in an environment that is always fluctuating. It is also necessary to avoid manipulating the natural lens, which can induce rapid onset of a premature cataract.
In addition, children’s eyes have a much more aggressive inflammatory response than adult eyes. Any internal surgical manipulation can generate near-immediate inflammation, characterized by the production of fibrin, a sticky material involved in wound healing and immunologic defense.
“Within minutes of doing anything inside a young child’s eye, this dense, gelatinous fluid fills the space we’re working in,” says Dr. Goshe. “DMEK requires us to inject and unroll a layer of tissue in the watery environment of the cornea. It’s a very delicate maneuver. When we do it in a child, that watery environment becomes more like sludge due to the inflammatory response. It’s almost impossible to complete a DMEK in that type of situation.”
Advertisement
In 2019, Dr. Goshe was the first surgeon to report on the novel use of tissue plasminogen activator (tPA) to reduce fibrin response in the eyes of a 4-year-old patient having DMEK. Although intraocular tPA had been used for many years, it had not yet been used to offset pediatric fibrin formation during DMEK. At the time, the patient was the youngest reported in the medical literature to have successful DMEK. Findings from that case led Dr. Goshe to inject tPA prophylactically in the eye of the 1-year-old.
“When injecting tPA in adults as uveitis therapy, it takes about 10-15 minutes to mitigate the fibrin reaction,” he says. “During DMEK in the 1-year-old, after the eye was prepared, we injected tPA in the anterior chamber about 10-15 minutes before transplanting the new endothelium. This allowed us to more easily and completely position the new, healthy layer of cells.”
Watch highlights of this surgery in the video below.
Video content: This video is available to watch online.
View video online (https://www.youtube.com/embed/QxKKo9iWogM?feature=oembed)
DMEK in 1-Year-Old (Graphic)
Typically, after the new endothelium is positioned, a temporary gas bubble is injected into the anterior chamber to hold the graft in place. During their first days of healing, patients are instructed to lie on their back, keeping the bubble in position and not blocking fluid flow through the pupil. However, this protocol is not practical for a 1-year-old. As such, the surgical team created a solution to allow the patient to be more mobile.
“We made two tiny holes in the iris as safety valves,” says Dr. Goshe. “In adults, we usually make only one. Having two holes meant that even if the patient’s head was positioned improperly, it would be nearly impossible for the gas bubble to obstruct both safety valves concurrently. We wouldn’t need to worry as much about a dangerous increase in intraocular pressure.”
Advertisement
After surgery, the patient returned home, where for one week his parents attempted to keep him in supine position as much as possible. During a follow-up visit one week after surgery, the parents reported that their child was more visually interactive. He wasn’t holding objects as closely to his eyes. The color of his iris appeared more vivid, and his pupil was more distinct. He had begun walking more independently and moving more confidently. The parents believed the surgery had been successful — remarking on how quickly their child had improved — and wanted their child’s second eye to receive the same treatment.
“At this point, timing was even more critical,” says Dr. Goshe. “In kids, if you improve one of their two eyes, you create a comparison point. They will rapidly gravitate vision to their better eye, putting themselves at higher risk for amblyopia in the untreated eye.”
One month later, the surgical team performed an identical procedure on the patient’s second eye.
The patient will continue use of steroid eye drops, tapering over one year. He will be followed by a pediatric ophthalmologist, who will conduct periodic eye exams to monitor the child’s intraocular pressure and determine his need for corrective lenses. The patient’s long-term prognosis is extremely good. The parents report that their child is no longer bothered by bright light and acts “like a new kid.”
Longevity of the cornea transplant is highly variable, optimistically 10-15 years. DMEK can be repeated later, as needed, and will be easier as the patient gets older, notes Dr. Goshe.
“To date, this patient is among the youngest reported in the medical literature to have DMEK,” he says. “This case demonstrates the need to be extra vigilant in patients with risk factors for an ocular condition. Most ophthalmologists have seen a patient with PPCD, but the disease has a range of manifestations and presents more aggressively in younger patients. We must continue to find innovative techniques to manage this and other challenging conditions in children.”
Advertisement

Motion-tracking Brillouin microscopy pinpoints corneal weakness in the anterior stroma

Study reveals more about the pathophysiology of Salzmann’s nodular degeneration
Studies continue to indicate effectiveness and safety

Identifies weak spots in the cornea before shape change occurs

Researcher gives guidance for clinical trials

A look at emerging technology shaping retina surgery

A multitude of subspecialities offer versatility, variety

Therapies include steroid implants, immunomodulation and biologics