A new study illustrates why it’s important for clinicians to be aware and suspicious of tip of the J-pouch leaks
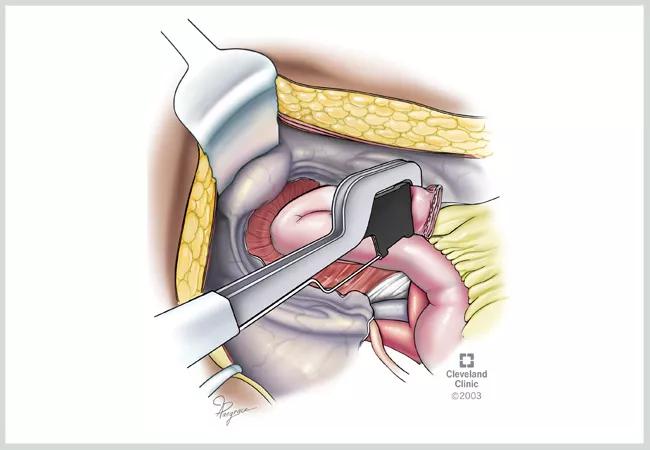
Figure 1. Stapled closure of the tip of the J-pouch. A technique felt by many to prevent this complication is to staple the tip of the J at an acute angle to improve the perfusion (blood supply) to the tip of the J which is the most distal part of the new J-pouch. This technique may also be used to make a new tip of the J-pouch during a pouch revision for a tip of the J leak. After the stapled tip of J construction, the staple line is often oversewn, again to prevent a leak (not shown). ©Cleveland Clinic, 2023
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
A recently published study1 from Diseases of the Colon & Rectum expands on a decades-old study2 that concluded that higher awareness of the possibility of a leak from the tip of the J-pouch — an uncommon complication of ileal pouch-anal anastomosis (IPAA) construction — could reduce pouch failure rates. The new study includes a larger cohort tracked over a longer period of time, and it reports similar findings as the original one. The authors hope the study can be used to increase education among caregivers regarding this rare but potentially devastating complication.
“The tip of the J-pouch is the farthest area from the aorta, and it’s the distal most aspect of the blood supply,” explains Stefan Holubar, MD, MS, IBD Surgery Section Chief and Director of Research in Cleveland Clinic’s Digestive Disease and Surgery Institute and lead author of the study. “It’s at the cut edge of the mesentery. We think leaks may potentially be an ischemic phenomenon i.e. related to low blood flow to that tip, but that’s not really been proven. This is a hypothesis, but it has biologic plausibility, and surgeons often take steps to try to prevent this. At Cleveland Clinic we typically transect the tip at an acute angle to improve blood supply to the anti-mesenteric tip.”
The study looked at 74 patients with leaks from the tip of the J-pouch (mean age 38 years old, 36 [48.6%] female and median BMI of 24.8 [20.7-27.2]). A total of 36 (48.7%) of the index IPAA operations were performed at Cleveland Clinic, with the others (n=38) being performed at referring institutions and subsequently transferred to Cleveland Clinic for further work-up and management.
Advertisement
The authors tracked the patient’s presenting symptoms and noted that pain was the most common (51 patients [68.9%]). The location of the pain included abdominal pain (35 patients [68.6%]), pelvis pain (eight patients [15.7%]), low back/coccygeal pain (six patients [11.8%]) and rectal/anal pain (three patients [5.9%]). Other common initial presenting complaints included fevers (32.4%), diarrhea and urgency (12.2%), nausea and vomiting (10.8%) and symptoms associated with fistulization to the bladder, vagina or skin (10.8%). Most of the cohort had multiple presenting symptoms, though four patients were asymptomatic.
A total of 55 (74.3%) patients had the leak of the tip of the J-pouch found using diagnostic testing, while 19 (25.6%) were only diagnosed intra-operatively. Some leaks (63.5%) were diagnosed only after loop ileostomy closure, while 32.4% were before ileostomy closure. The initial tests used to diagnose the leaks were pouchoscopy, pouchograms, contrast-enhanced computed tomography, fluoroscopic tube injection study, magnetic resonance imaging and fistulogram. Pouchoscopy (31.1%) and gastrografin enema (28.4%) were the most common methods used for diagnosis.
“Sometimes this is a delayed complication, occurring remote from the pouch construction and ileostomy closure, and this is probably because it was a contained leak or contained perforation that scarred and sealed,” explains Dr. Holubar. “This is best illustrated by the results of the study showing that a couple of the patients with the delayed presentations actually had peritonitis and the leak of the tip of the J-pouch leak was only discovered at the time of an emergency exploratory laparotomy for peritonitis.”
Advertisement
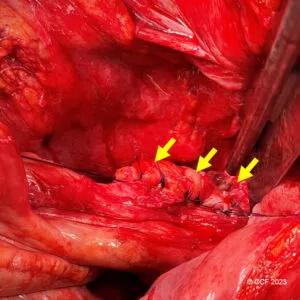
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/2aef74dd-818c-4998-af29-975dde6cf2cf/22-DDI-3503473-Tip-of-the-J-pouch-Leaks-Diagnosis-Management-inset1-300x300_jpg)
Figure 2. Sutured repair of a tip of the J leak with oversewing (inverting) the entire staple-line as shown by the arrows. ©Cleveland Clinic, 2023
A definitive non-operative approach was attempted in 48.6% but was successful in only 10.8% of patients overall. Initial surgical repair was attempted in 89.2% of patients, and only 4.5% required pouch excision. Salvage operations (n=63) included sutured or stapled repair of the tip of the J (65%, Figure 2), pouch excision with neo-pouch (25.4%), and pouch disconnection, repair and re-anastomosis (9.5%). Ultimately, 10 (13.5%) patients required pouch excision, yielding an overall five-year pouch survival rate of 86.3%.
“It was a little surprising to see the degree of heterogeneity of both the presentations and the management,” says Dr. Holubar. “Some of them are happening early while the patient is still diverted, and those are easier to manage and have less acute or less severe presentations. Others, presenting later usually have more advanced pelvic sepsis, which makes it harder to salvage the situation. They often require more aggressive or more intense surgical measures.”
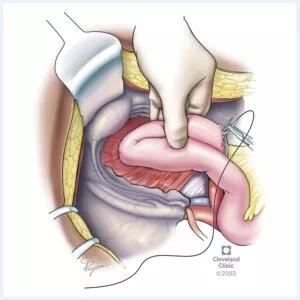
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/eac97934-4857-4089-939d-61de6497352e/22-DDI-3503473-Tip-of-the-J-pouch-Leaks-Diagnosis-Management-inset2-300x300_jpg)
Figure 3. Sutured closure of a leak from the tip of the J-pouch. The other method of local repair for tip of the J-pouch leaks is sutured closure. ©Cleveland Clinic, 2023
Dr. Holubar notes that, in general, complications of the pouch typically present themselves within the first couple of years, though not always. His team performed a study a couple of years ago looking at conditional pouch survival.3 They found that if the patient makes it through the first couple of years of a pouch without a complication, then the pouch survival actually goes up since the majority of complications with a pouch occur in the first year or first couple of years.
Advertisement
“I think the earlier we can treat something, the higher the success rate’s going to be,” explains Dr. Holubar. “This is especially true if the patients are still diverted. A couple of the patients that had tip of the J leak diagnosed early while still diverted had very minor leaks that were only treated with antibiotics and keeping the ileostomy for a couple of extra months and the leaks basically healed which is great.”
At its core, Dr. Holubar believes tip of the J-pouch leaks are part of a larger national quality issue. About 50% of the pouch surgeries in the United States are done at about 10 hospitals, which means the other 50% of the pouches in the United States are performed amongst all the other hospitals.
“In the surgical oncology world, this issue of volume and quality for pancreatic cancer has been dealt with in Whipple procedure volumes for example, but for benign disease, like pouches, it has not yet been widely adopted,” says Dr. Holubar. “There are several reasons for this — surgeons like to operate and that’s how they make their living, and they don’t want to give up a great case like a pouch. But the fact of the matter is that we know that there’s a correlation between high-volume surgery and improved outcomes. The American Society of Colorectal Surgery is working on trying to increase the exposure that colorectal fellows get to J-pouches, which is admirable. But the question still remains — if you see five pouches during your one-year fellowship, is that really adequate training to then go out and treat patients as a low-volume pouch surgeon? That’s a difficult question, and it’s one that our healthcare system is grappling with right now. I often get asked how many pouches per year makes a high-volume surgeon, but the real question is how many are too few. Most would agree one or two pouches a year is just not enough.”
Advertisement
Reference
Advertisement

Retrospective analysis looks at data from more than 5000 patients across 40 years

Radiation to the pelvis from cancer treatment made the traditional treatment path unavailable

A proof-of-concept study shows how 3D pouchography can be a useful adjunct for surgeons
Three presentations from the DDW 2023 conference help identify best candidates and illustrate the positive impact redo IPAAs can have on a patient’s QoL

Benefits of neoadjuvant immunotherapy reflect emerging standard of care

Multidisciplinary framework ensures safe weight loss, prevents sarcopenia and enhances adherence

Study reveals key differences between antibiotics, but treatment decisions should still consider patient factors

Key points highlight the critical role of surveillance, as well as opportunities for further advancement in genetic counseling