Case report demonstrates utility in a brain-injured patient
By Brittany Cunningham, PharmD; Amanda Staudt, APRN; Amy Renner, APRN; Gregory Griffin, DO; and Christopher R. Newey, DO, MS
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
After neurological injury, the brain is susceptible to secondary injury from processes that increase metabolic demand, such as fever.1,2 For every 1oC increase in temperature, cerebral metabolic demand increases by 8%.3 Fever is associated with increased morbidity and mortality in patients with brain injury from multiple etiologies.1,4 The Copenhagen Stroke Study found that there was a 30% increase in mortality for every 1°C above 37°C in patients with acute ischemic stroke.5 Fevers also contribute to increased length of stay in the neurologic ICU.6
The duration of targeted temperature management varies by clinical situation. The current recommendation for out-of-hospital cardiac arrest temperature management is a minimum of 24 hours.1 This duration maybe longer in other brain injuries.1 Multiple methods can be used for temperature management. The most reliable and effective means to cool is via conduction, which is the direct transfer of heat between surfaces where the amount of heat loss is related to contact surface.1
One such conductive method is the use of a gel pad surface cooling device, which consists of hydrogel-coated pads that adhere to the patient’s midsection and thighs. Temperature-controlled water is circulated under negative pressure to maintain the core body temperature at a predetermined temperature. We report here the case of a hospitalized patient with brain injury who developed a fever requiring gel surface cooling. We maintained his temperature within a targeted range using the novel maintenance mode on a recently updated gel pad surface cooling device.
Advertisement
A 79-year-old man with a history of diabetes, hyperlipidemia and paroxysmal atrial fibrillation was admitted to our hospital with a headache and gait instability. He was taking apixaban 5 mg twice daily. He reported having fallen two weeks prior to admission.
Head CT showed a right convexity subdural hemorrhage measuring 1.2 cm with 2 mm of midline shift (Figure 1A). INR was 1.18 (normal, 0.9-1.30), partial thromboplastin time was 28.8 seconds (normal, 23.0-32.4) and platelet count was 260,000/mm3 (normal, 141,000-365,000). He was given 4,500 units of activated prothrombin complex concentrate (50 U/kg). Repeat head CT was stable. Neurologically, he had no focal deficits.
On hospital day 4, he was taken to the operating room for subdural hemorrhage evacuation. In the immediate postoperative period he was found to have increasing encephalopathy and left hemiparesis. Repeat CT head showed reaccumulation of subdural hemorrhage with 8 mm of midline shift (Figure 1B). Repeat coagulation studies and platelets again were normal.
He was taken emergently to the operating room for a second evacuation, during which he was noted to have extensive oozing from the underside of the dura. Postoperative head CT again showed reaccumulation of subdural hemorrhage with midline shift (Figure 1C). He was given fresh frozen plasma and remained comatose. Coagulation studies and platelets remained normal. Fibrinogen was 798 mg/dL (normal, 200-400), and antithrombin activity assay was 97.6% (normal, 84.0%-123.0%).
Advertisement
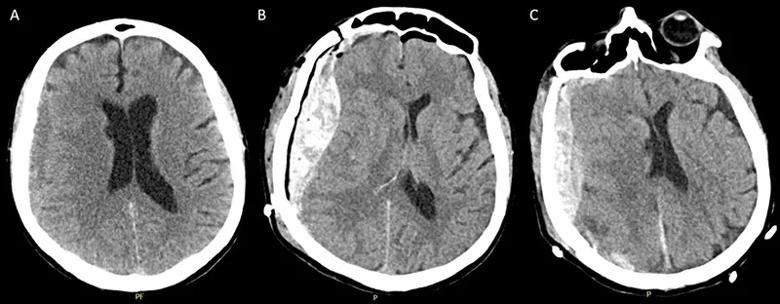
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/c293a652-6ec9-47a7-985f-fc980d4dfb1c/20-NEU-2018222-Case-study_Gel-pad-surface_inset-Figure-1_800x312_jpg)
Figure 1. Head CTs at presentation, showing the right convexity subdural hemorrhage with thickness of ~1.2 cm (A), immediately after the initial subdural hemorrhage evacuation (B) and following the second evacuation (C).
On hospital day 5, he developed status epilepticus and required treatment with midazolam infusion, fosphenytoin and levetiracetam. On hospital day 6, he developed a fever of 43oC. Cultures were obtained, and he was started on antibiotics. His temperature remained elevated.
He was placed on a gel pad surface cooling device using monitor mode, with a set temperature range of 36° to 37.5°C (Figure 2). He remained on this monitor mode for four days, at which point the setting was changed to normothermia mode with a set temperature of 37°C (red arrow). During the monitor mode, the water temperature of the gel pad surface cooling device remained fairly continuous and smooth compared with the normothermia mode (after the red arrow). He remained clinically unchanged and was transitioned to hospice care.
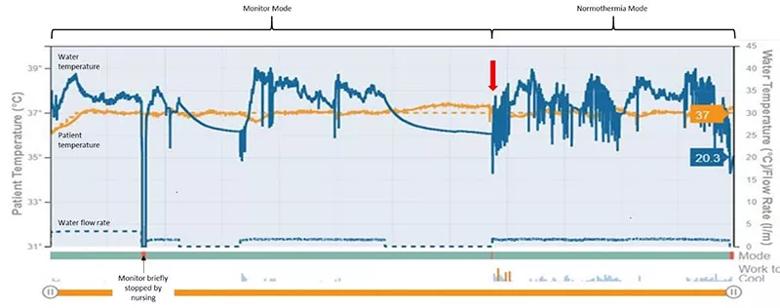
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/51b1d365-e531-4134-9d44-4877053dd631/20-NEU-2018222-Case-study_Gel-pad-surface_Inset_Figure-2_800x316_jpg)
Figure 2. Temperature tracings over time from the patient’s gel pad surface cooling device. The device’s monitoring mode was used for four days before the setting was transitioned to normothermia mode (37°C; red arrow). During the use of monitoring mode, the patient’s temperature was kept between 36° and 37.5°C with a more consistent water temperature relative to the post-monitor mode period (i.e., after the red arrow). Blue line indicates the device’s water temperature. Yellow line indicates patient temperature.
Advertisement
This case is, to our knowledge, the first reported use of the novel monitor mode on the updated gel pad surface cooling device. This mode allows the user to set upper and lower temperature limits. It also allows the user to set a length of time during which a patient can be outside these limits. By permitting the setting of a temperature range, the new monitor mode allows for improved patient comfort. A range will reduce the water temperature fluctuations and allow for a more consistent and stable water temperature, as we have demonstrated in this case.
Advertisement
Dr. Newey is Medical Director of the Neurosciences ICU at Cleveland Clinic Akron General and Medical Director of Multimodal Monitoring for Cleveland Clinic. All other co-authors are clinical providers at Cleveland Clinic Akron General.
Advertisement

An argument for clarifying the nomenclature

An expert talks through the benefits, limits and unresolved questions of an evolving technology

Recommendations on identifying and managing neurodevelopmental and related challenges

Phase 2 trials investigate sitagliptin and methimazole as adjuvant therapies

Aim is for use with clinician oversight to make screening safer and more efficient

Rapid innovation is shaping the deep brain stimulation landscape

Study shows short-term behavioral training can yield objective and subjective gains

How we’re efficiently educating patients and care partners about treatment goals, logistics, risks and benefits