Cleveland Clinic research team uses fMRI to assess real-time brain activity
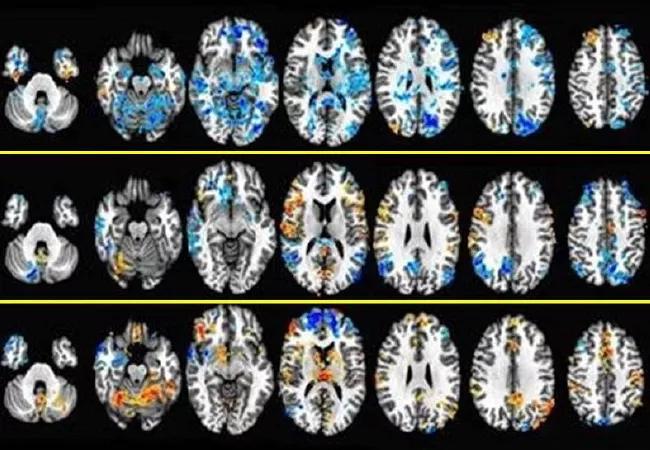
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/3576956c-be52-4db2-9346-13d6af67ab2e/650x450-fMRI-Study_jpg)
650×450-fMRI-Study
By Howard B. Goldman, MD; Bradley Gill, MD, MS; Javier Pizarro-Berdichevsky, MD; Pallab Bhattacharyya, PhD; Sandip Vasavada, MD; and Stephen Jones, MD, PhD
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
Voiding involves a cascade of complex neurological processes. In overactive bladder, one or more of those processes may go awry.
Sacral neuromodulation (SNM) therapy has proven effective in treating the wayward neurological processes for many patients with OAB. Improving our understanding of the mechanism of action of SNM could help improve the selection of patients who might benefit from this treatment and potentially allow for improvements in the technology.
To this end, a multidisciplinary Cleveland Clinic research team conducted the first-ever study using functional magnetic resonance imaging (fMRI) to measure brain activity in women with OAB who had success using SNM. Results of this study are published in The Journal of Urology.
Our investigation was a culmination of years of work, including several safety studies needed to garner Institutional Review Board approval for enrolling patients. Generally, fMRIs are not allowed for patients with implanted electrical devices.
Thirteen female patients, median age 52 (age range 36 to 64), were enrolled in our study. Six completed the fMRI imaging portion of the study. All completed a voiding diary. Mean baseline daytime frequency in this cohort was 5.8 + 1.7 voids, 0.83 + 1.6 nocturia episodes and 0.17 + 0.41 daily episodes of urgency urinary incontinence on SNM.
For the study, we turned off patients’ SNM devices for one week, causing their symptoms to return. Then we performed fMRI on each patient while we turned the SNM on and off. Patients underwent three fMRI sessions with their devices operating at different levels of intensity each time: subsensory, sensory and suprasensory.
Advertisement
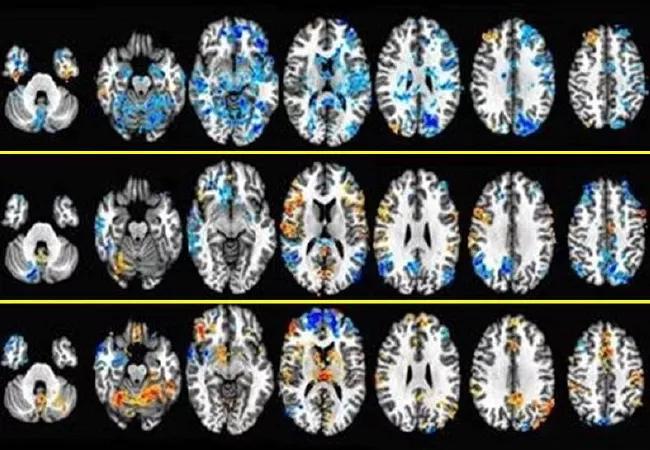
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/3576956c-be52-4db2-9346-13d6af67ab2e/650x450-fMRI-Study_jpg)
FMRI images demonstrating areas of deactivation (bluer) and activation (redder) in the brain at different intensities of stimulation.
The data yielded some interesting results. In general, brain activation progressed with increased stimulation levels. However, activation of the inferior frontal gyrus remained stable, and deactivation of the pons and the periaqueductal gray matter only occurred with subsensory stimulation. Sensory stimulation activated the insula, which previous studies have shown is activated upon bladder filling, but deactivated the medial and superior parietal lobes. Suprasensory stimulation activated multiple structures, including sensory regions of the S3 nerve root where the stimulator lead is implanted.
The mechanism of action in SNM has long been hypothesized to involve influencing neural circuits in the peripheral and central nervous system (CNS). This study showed real-time changes in brain activity with SNM therapy, supporting the hypothesis that SNM works in part by influencing pathways in the CNS. We also observed that altered stimulus intensity elicited an amplitude-response relationship in various brain regions.
Our challenge now is to further clarify how changes in brain activity from SNM affect urinary tract function. Does SNM provide a modulatory signal that alters the set point in brain centers that coordinate lower urinary tract function and voiding – like a thermostat? In OAB, this set point is likely miscalibrated.
Or does SNM produce afferent activity that alters aberrant neural activity underlying OAB? An analogy for this is how noise-cancellation headphones enable soft music to be heard in a noisy environment.
Advertisement
Our study achieved its aim of showing that SNM devices alter brain activity. In the future, we hope to change the parameters used in fMRI testing, such as performing fMRI when patients have a full bladder or with different stimulus settings, to tease out more details on how SNM devices affect neural structures and circuits.
Dr. Goldman has joint appointments in the departments of Urology and Obstetrics/Gynecology. He is a consultant for and received research support from Medtronic for this study. Dr. Gill is a resident and Dr. Pizarro-Berdichevsky was a clinical fellow in the Glickman Urological & Kidney Institute. Dr. Bhattacharyya is scientist in Cleveland Clinic’s Mellen Center for Multiple Sclerosis. Dr. Vasavada is Urologic Director, Center for Female Urology and Reconstructive Pelvic Surgery. Dr. Jones is in the Diagnostic Radiology Department. Katherine Koenig, PhD, assisted with image processing and fMRI data analysis.
Advertisement
Advertisement
Study finds broadly similar outcomes between MIS and open surgical approaches
System uses clinical data routinely collected at clinical visits
Newer protocols address testing, language and communication
Collaboration with AI startup promises to reshape neurocritical care monitoring at scale
Study looked at mobility measures and safety
Types and presentation may differ from adults, but early recognition and intervention are just as key
Study tests potential for a more accurate treatment predictor
Study uses molecular and clinical stratification to help guide patient selection