Severity of diabetic retinopathy is a useful predictor
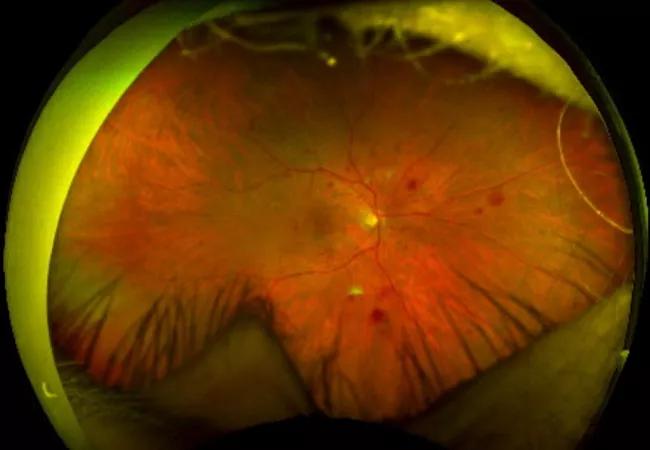
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/6afed46e-91eb-4b7f-9f2a-2ccbf63280ad/21-EYE-2085257-CQD-Diabetic-Retinopathy-Hero_jpg)
21-EYE-2085257-CQD-Diabetic-Retinopathy-Hero
By Katherine E. Talcott, MD; Carolina C.S. Valentim, MD; and Rishi P. Singh, MD
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
The most common cause of vision loss among patients with diabetic retinopathy (DR) is diabetic macular edema (DME). DME can develop at any stage of DR. Intravitreal anti-vascular endothelial growth factor (anti-VEGF) agents have become the mainstay of treatment for DME, but clinical response varies greatly. Some patients show improvement in visual acuity and on optical coherence tomography (OCT) after a few weeks of treatment while others need repeat injections or never achieve complete resolution of edema.
Recognizing predictive factors in treatment response is imperative for optimizing management strategies, which can decrease the risk of complications and reduce financial burden for patients and providers.
Our research team at Cole Eye Institute studied how baseline DR severity corresponded with DME resolution in patients treated with anti-VEGF in two large phase 3 clinical trials. Baseline predictors of time to DME resolution also were evaluated.
DR severity can be assessed with a DR severity scale (DRSS) based on intraretinal microvascular abnormalities, hemorrhages, microaneurysms and venous beading, among other findings on fundus photographs. For our study, baseline DR severity was categorized in three groups:
Our team performed a post hoc analysis of patients treated with ranibizumab, an intravitreal anti-VEGF agent, in the phase 3 RIDE (NCT00473382) and RISE (NCT00473330) clinical trials. In RIDE and RISE, 759 patients with DME were randomized to two doses (0.3 mg or 0.5 mg) of monthly ranibizumab or sham for a total of 24 months. Our analysis included 416 ranibizumab-treated patients (0.3 mg and 0.5 mg doses pooled) with significant edema on OCT images (central subfield thickness [CST] ≤ 250 µm) and diabetic retinopathy at baseline. DRSS scores ranged from 35 (mild nonproliferative DR) to 85 (advanced proliferative DR).
Advertisement
Resolution of DME was assessed at day 7 and months 1, 2, 3, 6, 12, 18 and 24 of the trial. DME resolution was defined as the first visit with CST ≤ 250 µm on OCT imaging. The frequency of DME resolution at months 12 and 24 was assessed by baseline DR severity. Mean time to DME resolution was also assessed by baseline DR severity. Finally, baseline factors associated with time to DME resolution were evaluated.
We found that the frequency of DME resolution across baseline DR severity subgroups (DRSS scores 35/43, 47-53 and 60-85) was similar at month 12 (52.6%, 55.0% and 54.6% respectively) and month 24 (70.7%, 68.9% and 65.9% respectively). The mean time to first DME resolution was 8.6 months for patients with mild and moderate nonproliferative DR (baseline DRSS scores 35/43), 8.0 months for those with moderately severe and severe nonproliferative DR (baseline DRSS scores 47-53), and 5.8 months for those with advanced proliferative DR (baseline DRSS scores 60-85). There was a significant difference between these groups (P = 0.03).
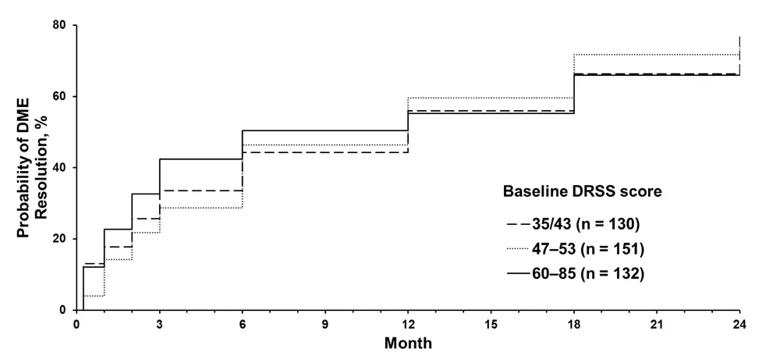
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/f2d70503-70d8-4398-91fa-b70e8e2a54d0/21-EYE-2085257-CQD-Diabetic-Retinopathy-Inset_jpg)
Kaplan-Meier plot of time to DME resolution over 24 months, stratified by baseline diabetic retinopathy severity.
Additionally, we found the rate of DME resolution by month 24 was significantly higher in patients who had not had cataract surgery versus those who had cataract surgery at baseline (72.1% vs. 59.8%; P = 0.01). No other baseline variables were significantly associated with time to DME resolution.
Based on the results of this study, our group concluded that the proportion of patients treated with anti-VEGF who achieved DME resolution at months 12 and 24 of RIDE and RISE was similar across baseline DR severity subgroups. However, there was a trend for shorter time to DME resolution in patients with more severe DR at baseline.
Advertisement
Our group was surprised by these results as we expected those with less severe retinopathy levels at baseline to have the fastest times to dryness due to milder diabetic eye disease. Conversely, those with the most severe retinopathy levels had the fastest times to dryness. Our results indicate that DME in eyes with more severe DR may be more driven by VEGF rather than inflammatory mediations and are more responsive to anti-VEGF treatment.
These results have helped improve our understanding of DME pathophysiology, which might not be the same for all levels of DR severity. This work allows clinicians to better forecast how patients will progress with treatment and may help when selecting therapies.
Dr. Talcott is a principal investigator and Dr. Valentim is a research fellow in the Center for Ophthalmic Bioinformatics at Cleveland Clinic’s Cole Eye Institute. Dr. Singh is Director of the center.
Advertisement
Advertisement
The drug does not provide lasting disease control for most patients
New service for patients with diabetes will make annual screening more convenient
Gonioscopy can help determine next steps in reducing IOP
Similarly, patients with noninfectious uveitis have a higher risk of being diagnosed with an autoimmune disease within five years
A review of pathophysiology, risk factors, clinical presentation and treatment options
How computational tools and personalized biomechanics can improve keratoconus detection, ectasia risk assessment and surgical outcomes
Motion-tracking Brillouin microscopy pinpoints corneal weakness in the anterior stroma
Registry data highlight visual gains in patients with legal blindness