The disease’s neuropathologic heterogeneity holds clues to refining diagnosis and prognosis
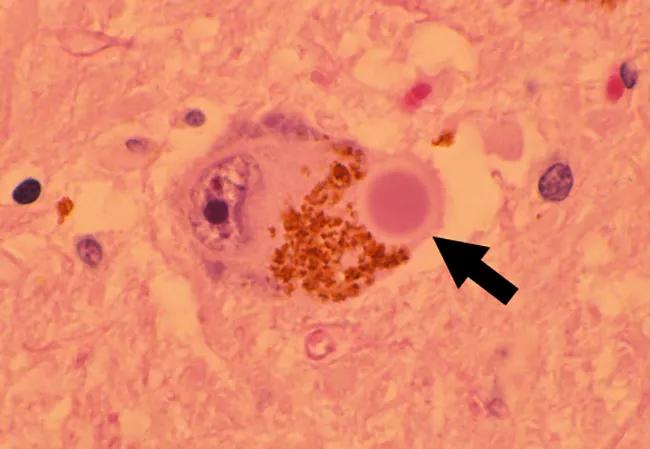
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/1e61e643-4dfc-4177-ad3e-6bdb31050973/23-NEU-3813860_lewy-body-inclusion_650x450-jpg)
histopathology image with pink background and arrow pointing to round cell
Dementia with Lewy bodies (DLB) is traditionally considered to be clinically and pathologically distinct from Alzheimer’s disease (AD). DLB is notably characterized by early executive and attention deficits, vivid visual hallucinations, REM sleep disorder and Parkinson-like movements, as well as the presence of Lewy bodies in the brain. In contrast, the typical initially episodic memory loss of AD is accompanied by amyloid plaques and tau neurofibrillary tangles.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
But this picture is anything but clear-cut. Not only do symptoms overlap, but studies have found that most people clinically diagnosed with DLB have not only Lewy bodies but also pathologic features of AD.
Two recently published research articles shed light on this confusing situation. One (Neurology. 2025;105[12]:e214346) is a cross-sectional cohort study that found that just 68% of clinically diagnosed DLB patients tested positive for the CSF α-synuclein seed amplification assay (αSyn-SAA+) — which detects the misfolded protein specific to Lewy bodies and Lewy neurites — and that these patients were likelier to have a rapid disease course than those who tested negative (αSyn-SAA–).
The second (Neurol Open Access. 2026;2:e000055), a retrospective longitudinal cohort study, examined behavioral and psychological symptoms of dementia, which tend to be overlooked compared with cognitive symptoms in describing both DLB and AD. Incorporating such symptoms, the investigators defined three distinct clinical phenotypes cutting across DLB, AD and mixed AD-DLB, enabling prediction of which patients would experience a more rapid versus slower decline.
“Findings from these studies can enhance the evaluation of patients with DLB,” says Cleveland Clinic behavioral neurologist Jagan Pillai, MD, PhD. “This has important implications for improving diagnosis and prognosis and for refining cohorts to optimize clinical research.”
Dr. Pillai was principal and corresponding author of the study analyzing behavioral features and contributed to the article focused on αSyn-SAA. He is Director of the Cleveland Alzheimer’s Disease Research Center, which recently received a $4.9 million grant from the National Institutes of Health to fund research on characterizing cognitive fluctuations — a core feature of DLB but also common in AD — which may provide clues to understanding the underpinnings of dementia.
Advertisement
For the research focused on the αSyn-SAA, the cohort came from the DLB Consortium, part of the National Institute of Neurological Disorders and Stroke (NINDS) Parkinson’s Disease Biomarkers Program. Their registry includes a well-characterized population clinically diagnosed with DLB at tertiary care centers in the U.S.
Among 99 participants with available data on CSF αSyn-SAA and AD biomarkers, the following results were found:
Dr. Pillai notes that the αSyn-SAA– group was heterogeneous and possibly includes patients with primary AD, an atypical parkinsonian syndrome or vascular dementia. Autopsy evidence would be needed to determine this.
“Patterns of αSyn-SAA results and the presence of AD biomarkers reveal that patients diagnosed with DLB likely vary in their underlying pathology,” Dr. Pillai explains. “The patterns provide clues to better define cohorts and predict disease course.”
The second study’s cohort included 1,028 patients from the National Alzheimer Coordinating Center with autopsy confirmation of either AD (n = 521), DLB (n = 96) or mixed AD-DLB (n = 411). All had a baseline Clinical Dementia Rating Global score of 1 or less (mild to no dementia). In addition to standard cognitive testing, the patients had been evaluated with the Neuropsychiatric Inventory Questionnaire, which assesses for multiple behavioral and psychological symptoms (agitation, apathy, depression, delusions, disinhibition, auditory and visual hallucinations, irritability, personality change and REM sleep disorder).
Advertisement
Based on cognitive profiles and behavioral and psychological symptoms at very early stages of functional impairment, the investigators identified three clinical subtypes across the three pathology-based diagnoses and evaluated the differences in clinical outcomes among the groups.
Among patients with DLB, the fastest cognitive and functional decline occurred in those with the following characteristics at baseline:
On the other hand, patients who progressed more slowly in the AD and mixed AD-DLB groups generally were older, had better memory and were less likely to be an APOE Ɛ4 carrier.
“Despite the neuropathological heterogeneity among patients with dementia, early cognitive and behavioral symptoms provide important clues to predicting their prognosis,” Dr. Pillai notes.
“Both studies have identifiable subgroups of people who progress faster,” Dr. Pillai says. “This can have critical impacts in research and patient care.”
The implications for clinical care are many. Patients and their families can be given better information about the course their disease is likely to take. At the same time, physicians can better determine the need for additional testing, especially for patients with a mismatch between biomarkers and clinical presentation. Finally, physicians can steer clinical support systems toward patients who are most likely to need them early on.
These findings may also lead to smarter clinical trial design. “Better ability to distinguish among different groups allows us to create more uniform cohorts,” Dr. Pillai concludes. “This makes it more likely that we will detect signals of treatment benefit, potentially enhancing development of more-targeted therapies.”
Advertisement
At top: Pathology image indicating a Lewy body.
Advertisement
Advertisement
Study provides Class III evidence that assay distinguishes disease from normal controls
Will enable sharper focus on new diagnostic tools for elusive neurodegenerative disease
A population with very high lifetime risk presents care challenges and pathophysiologic insights
Two Cleveland Clinic neurologists review biomarker advances, targeted therapies and unresolved clinical challenges
GenT framework aims to improve drug development with focus on entire genes, not individual mutations
New research highlights serious risks and the critical need for earlier advance care planning
An expert talks through the benefits, limits and unresolved questions of an evolving technology
How we’re efficiently educating patients and care partners about treatment goals, logistics, risks and benefits