Does tumor location matter?
Adjunctive therapy appears to benefit only pancreatic head tumors and not body and tail tumors after surgical resection, a new study by Cleveland Clinic investigators suggests. Davendra Sohal, MD, MPH, staff in the Department of Hematology and Medical Oncology, collaborated with colleagues including Alok Khorana, MD, FASCO, Director, Gastrointestinal Malignancies Program, to evaluate a potential relationship between tumor site and benefit from adjuvant, neoadjuvant and perioperative therapies for pancreatic cancer. Results appear in Surgical Oncology.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
Multi-agent chemotherapy regimens are one of the few developments supporting improved overall survival (OS) in pancreatic adenocarcinoma. Oncologists are beginning to agree that early systemic therapy can improve outcomes, but most evidence supporting adjunctive therapy for resected pancreatic cancer focuses on head tumors. “We wanted to know if tumor location matters,” says Dr. Sohal, “so that we can better pinpoint which patients benefit from adjunctive therapies.”
The team completed a comprehensive analysis of patients (N = 27,930) with resected pancreatic adenocarcinoma in the National Cancer Database. The study population included patients diagnosed with stage I and II pancreatic cancer between 2003 and 2013 who underwent resection of the pancreatic primary and whose records mentioned tumor site (head, body or tail).
The median age of patients was 66 years; 51 percent were male, and 86 percent were white. Most of the tumors were head (74.4 percent), followed by tail (16.3 percent) and body (9.3 percent). More than three-fourths of cancers were stage II, and 81 percent had negative margins. Median OS was 24 months (21.6 for head, 34.5 for body and 42.5 for tail).
In univariable testing, the use of adjunctive therapy was associated with worse OS for tail and body tumors. Multivariable analysis confirmed a clear interaction between the site of the primary tumor and treatment. Multivariable models separated by tumor site showed a significant association between treatment and pathologic stage for head tumors but not for body and tail. The benefit was greatest with perioperative therapy. Another univariable analysis showed that adjunctive therapy was only beneficial for clinical stage II head tumors.
Advertisement
“Our analysis also showed that tail and body tumors are associated with significantly better survival,” says Dr. Khorana. “We of course need prospective studies to further examine this association, but our study offers the most statistically robust evidence to date.”
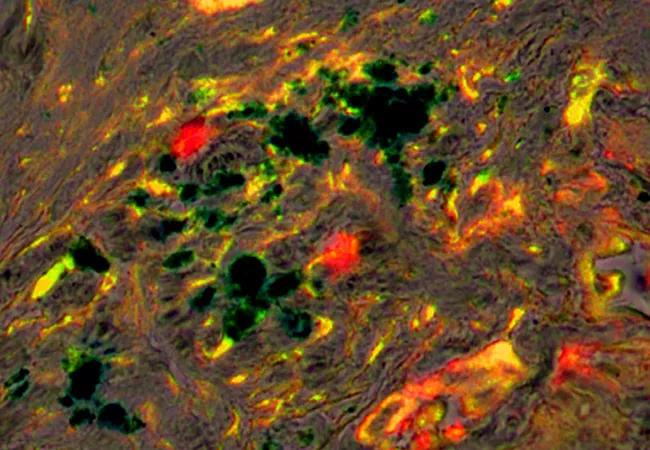
Image: This image shows magnetic iron nanoparticles that target cells with IGF-1R receptors, and are conjugated to a chemotherapy drug (dark blue). In the tumor stroma of a mouse model of pancreatic cancer, the nanoparticles delivered the chemotherapy to tumor-associated macrophages expressing IGF-1R (red) and CD68 (green). Source: National Cancer Institute.
Advertisement
Advertisement

Combining advanced imaging with targeted therapy in prostate cancer and neuroendocrine tumors

Early results show strong clinical benefit rates

The shifting role of cell therapy and steroids in the relapsed/refractory setting

Radiation therapy helped shrink hand nodules and improve functionality

Standard of care is linked to better outcomes, but disease recurrence and other risk factors often drive alternative approaches

Phase 1 study demonstrates immune response in three quarters of patients with triple-negative breast cancer

Multidisciplinary teams bring pathological and clinical expertise

Genetic variants exist irrespective of family history or other contributing factors