Cleveland Clinic specialists author a review

Editor’s note: This is an abridged version of an article originally published in the Cleveland Clinic Journal of Medicine. This is the second article in a two-part series; a review of clinical presentations and diagnosis of pattern hair loss can be found here.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
By Nina L. Tamashunas, BS, and Wilma F. Bergfeld, MD, FAAD
The goal of treatment of pattern hair loss is to promote regrowth of hair to improve scalp coverage and to prevent or slow further hair thinning and loss. Topical minoxidil and oral finasteride are the first-line treatments for male pattern hair loss, and topical minoxidil is the first-line treatment for female pattern hair loss. However, there are a number of alternatives. Detecting and treating comorbidities such as androgen excess and nutritional deficiencies is helpful in maintaining hair growth.
Mechanism of action. Through an unclear mechanism, minoxidil enhances hair growth by enlarging miniaturized hair follicles, extending anagen, and shortening telogen.23
US Food and Drug Administration (FDA) approval. Minoxidil is FDA-approved for treating both male and female pattern hair loss.
Administration. Topical minoxidil is available over the counter in 2% and 5% solutions and 5% foam in the United States. Patients should apply 1 mL of 5% solution or half a cap of 5% foam once a day directly to involved areas of the scalp (not the hair) when dry. Treatment should be assessed for efficacy after 1 year of use, but results may be observed sooner.
Efficacy in men. In a 48-week, double-blind, placebo-controlled, randomized trial, 5% minoxidil solution was superior to 2% solution in terms of patient perception of hair growth and treatment benefit, investigator perception of hair growth, and nonvellus hair count.24 Response time was also shorter with the 5% solution. Both the 2% and the 5% topical minoxidil solutions were superior to placebo.
Advertisement
Foam has not been directly compared with liquid solutions. Compared with placebo, 5% minoxidil foam increased the hair count and improved hair loss as assessed by the patient and physician.25
Individual response to treatment with minoxidil varies, and hair regrowth can be lost after stopping.
Efficacy in women. A meta-analysis found that topical minoxidil was effective and safe for treating female pattern hair loss, with no significant difference in efficacy and safety between different concentrations.26 Topical minoxidil is an effective treatment for women regardless of androgen status and age.10
Side effects. Minoxidil stimulates telogen follicles to enter anagen, so transient hair shedding may occur when initiating therapy. Contact dermatitis and hypertrichosis are common side effects. Compared with 2% topical minoxidil, 5% preparations are more likely to cause local pruritus and irritation.25
Mechanism of action. Finasteride, a competitive inhibitor of type II 5-alpha reductase, decreases production of dihydroxytestosterone. Oral finasteride reduces scalp dihydroxytestosterone levels by approximately 60% to 70%, depending on the dosage.26
FDA approval. Finasteride is FDA-approved for treating male pattern hair loss, but not female pattern hair loss.
Administration. Men with male pattern hair loss can be treated with finasteride 1 mg daily. Treatment should be assessed for efficacy after 1 year of use, but results may be observed sooner.
Efficacy in men. Finasteride has been shown to increase the hair count, physician-assessed hair coverage, and hair mass compared with placebo.27,28
Advertisement
A systematic review of the efficacy of finasteride in men with male pattern hair loss found that 5.6 patients need to be treated short-term, and 3.4 patients need to be treated long-term, for 1 patient to perceive an improvement.27 There was a 20% absolute increase in patient-perceived improvement in the short term and a 30% absolute increase in the long term.27 Longer treatment with finasteride promotes greater therapeutic success.
Similar to minoxidil, response to finasteride varies, and hair regrowth can be lost after the medication is discontinued.
Efficacy in women. A double-blind, placebo-controlled, randomized multicenter trial found finasteride 1 mg to be ineffective in postmenopausal women with female pattern hair loss at 12 months.29 However, higher doses of finasteride daily can be effective in cases of female pattern hair loss associated with hyperandrogenemia.30
Side effects. In a systematic review, the only adverse effect associated with finasteride treatment was erectile dysfunction, with an absolute increase in risk of approximately 1.5%.27 Approximately 1 in every 80 men treated with finasteride experiences this side effect. Stopping finasteride generally leads to resolution of erectile dysfunction, but sexual dysfunction can persist in some patients.
Other reported side effects of finasteride include decreased libido, gynecomastia, testicular pain, and depression.
Considerations. Finasteride can result in lower prostate-specific antigen levels in men, which should be taken into consideration when interpreting laboratory results. Due to hepatic metabolism of finasteride, precautions should be taken with patients with liver disease. With chronic treatment, finasteride may cause mild to moderate elevations in serum liver enzymes, but this is usually self-limiting and rarely requires dose modification or drug discontinuation.31
Advertisement
Dutasteride, a potent type I and type II 5-alpha reductase inhibitor, is used to treat benign prostatic hyperplasia, but is also prescribed as an off-label treatment for pattern hair loss.
A randomized control trial32 in 416 men with male pattern hair loss demonstrated that dutasteride 2.5 mg was superior to finasteride 5 mg in terms of increasing hair count over 24 weeks. Additionally, a meta-analysis33 found that finasteride and dutasteride had similar efficacy in treating pattern hair loss.
Latanoprost is a prostaglandin F2 analogue that extends the anagen phase.34 A double-blind, placebo-controlled, randomized trial in 16 men demonstrated an increase in hair density when latanoprost 0.1% was applied to a small area of the scalp.35
Bimatoprost is also being investigated as a potential treatment for pattern hair loss.
Ketoconazole, an antifungal medication that has anti-inflammatory and antiandrogenetic properties, can be used to treat seborrheic dermatitis and dandruff.36 It was found to reduce inflammation in pattern hair loss and to benefit women with hyperandrogenemia and female pattern hair loss when used as a 2% shampoo.37
Over-the-counter zinc-containing shampoos have a similar mechanism of action and therapeutic effect.
Spironolactone competitively inhibits the androgen receptor and also inhibits ovarian production of androgens. Of the antiandrogen drugs used off-label, it is the one most commonly used. It has been used to treat female pattern hair loss for over two decades and has a good safety profile.37 An open-label trial38 showed that spironolactone 200 mg daily improved hair regrowth in 44% of patients at 12 months, which was comparable to cyproterone acetate.
Advertisement
Cyproterone acetate is a progesterone derivative that prevents dihydroxytestosterone from binding to the androgen receptor and inhibits release of follicle-stimulating hormone and luteinizing hormone, thereby reducing testosterone levels. It is available combined with ethynyl estradiol as an oral contraceptive in many countries but is not approved in the United States.
Platelet-rich plasma is an autologous preparation of plasma with platelets, growth factors, and cytokines. It was initially used during hair transplantation procedures, with mixed results. Recently its use by itself has been explored to treat pattern hair loss. Preliminary evidence suggests that platelet-rich plasma may be advantageous in hair regrowth.39 Side effects include redness and pain at injection site and pinpoint bleeding.
Low-level laser therapy is an FDA-approved treatment for pattern hair loss. The mechanism by which it improves hair loss is unclear, but it may stimulate follicular stem cells or keratinocytes, increase blood flow, promote mitosis, increase cell metabolism, and have anti-inflammatory effects.40 Two double-blind, sham device-controlled, randomized trials demonstrated that low-level laser therapy increased hair density over 24 to 26 weeks of treatment, although differences global improvement ratings were not significant in one of the trials.41,42
Rolling fine needles over the skin causes minor physical trauma to the stratum corneum that incites wound healing. A randomized controlled trial demonstrated promising results with microneedling as an adjuvant to drug treatment for pattern hair loss.43 Side effects of the procedure include pain and pinpoint bleeding.
Nonmedical options allow patients to manage the appearance of thinning hair. Scalp colorants, including powders, lotions, and hair sprays, can reduce the color contrast between the skin and hair, camouflaging the scalp. The scalp can also be covered with wigs, hair extensions, or hair pieces.
Follicles can be transplanted from an unaffected area of the scalp, commonly the occiput, where there are more than 40 follicular units/cm3, to affected areas to create a permanent improvement in hair coverage. Young patients or patients with vertex involvement are not ideal candidates for this procedure due to the high likelihood of continued progression.10 A large number of grafts, 1,000 to 2,000 follicles, can be transplanted in a single session. Additional transplantation sessions are generally scheduled at least 6 months apart, as it takes 5 to 6 months for results to be established.10
Pharmacologic treatment, such as oral finasteride, can be used in conjunction with hair transplantation before and after sessions to prevent hair miniaturization and loss of non-transplanted hair follicles.44
The article in its entirety, including a complete list of references, can be found here.
Advertisement
Family history may eclipse sun exposure in some cases
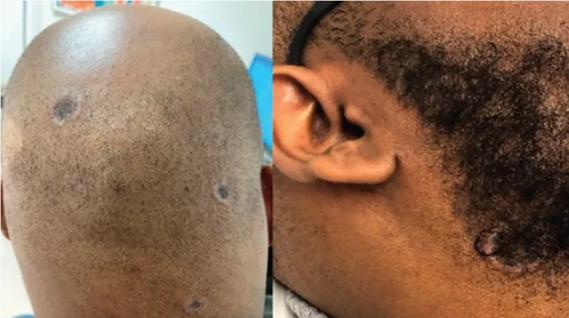
Consider secondary syphilis in the differential of annular lesions
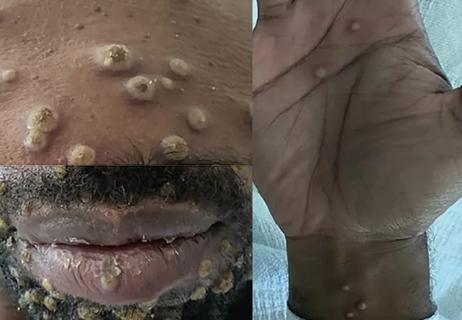
Persistent rectal pain leads to diffuse pustules

Two cases — both tremendously different in their level of complexity — illustrate the core principles of nasal reconstruction
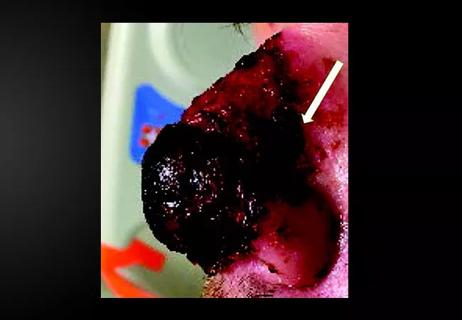
Stress and immunosuppression can trigger reactivation of latent virus

Low-dose, monitored prescription therapy demonstrates success

Antioxidants, barrier-enhancing agents can improve thinning hair